Differential orientation and conformation of surface-bound keratinocyte growth factor on (hydroxyethyl)methacrylate, (hydroxyethyl)methacrylate/methyl methacrylate, and (hydroxyethyl)methacrylate/methacrylic acid hydrogel copolymers
- PMID: 30360629
- PMCID: PMC6905655
- DOI: 10.1116/1.5051655
Differential orientation and conformation of surface-bound keratinocyte growth factor on (hydroxyethyl)methacrylate, (hydroxyethyl)methacrylate/methyl methacrylate, and (hydroxyethyl)methacrylate/methacrylic acid hydrogel copolymers
Abstract
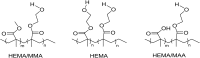
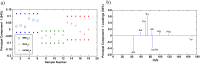
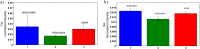
The development of hydrogels for protein delivery requires protein-hydrogel interactions that cause minimal disruption of the protein's biological activity. Biological activity can be influenced by factors such as orientational accessibility for receptor binding and conformational changes, and these factors can be influenced by the hydrogel surface chemistry. (Hydroxyethyl)methacrylate (HEMA) hydrogels are of interest as drug delivery vehicles for keratinocyte growth factor (KGF) which is known to promote re-epithelialization in wound healing. The authors report here the surface characterization of three different HEMA hydrogel copolymers and their effects on the orientation and conformation of surface-bound KGF. In this work, they characterize two copolymers in addition to HEMA alone and report how protein orientation and conformation is affected. The first copolymer incorporates methyl methacrylate (MMA), which is known to promote the adsorption of protein to its surface due to its hydrophobicity. The second copolymer incorporates methacrylic acid (MAA), which is known to promote the diffusion of protein into its surface due to its hydrophilicity. They find that KGF at the surface of the HEMA/MMA copolymer appears to be more orientationally accessible and conformationally active than KGF at the surface of the HEMA/MAA copolymer. They also report that KGF at the surface of the HEMA/MAA copolymer becomes conformationally unfolded, likely due to hydrogen bonding. KGF at the surface of these copolymers can be differentiated by Fourier-transform infrared-attenuated total reflectance spectroscopy and time-of-flight secondary ion mass spectrometry in conjunction with principal component analysis. The differences in KGF orientation and conformation between these copolymers may result in different biological responses in future cell-based experiments.
Figures
Similar articles
-
ToF-SIMS and TIRF microscopy investigation on the effects of HEMA copolymer surface chemistry on spatial localization, surface intensity, and release of fluorescently labeled keratinocyte growth factor.Biointerphases. 2019 Sep 23;14(5):051003. doi: 10.1116/1.5119871. Biointerphases. 2019. PMID: 31547664 Free PMC article.
-
Novel hydrogel membrane based on copoly(hydroxyethyl methacrylate/p-vinylbenzyl-poly(ethylene oxide)) for biomedical applications: properties and drug release characteristics.Macromol Biosci. 2005 Oct 20;5(10):983-92. doi: 10.1002/mabi.200500091. Macromol Biosci. 2005. PMID: 16208632
-
Novel porphyrin-incorporated hydrogels for photoactive intraocular lens biomaterials.J Phys Chem B. 2007 Jan 25;111(3):527-34. doi: 10.1021/jp066217i. J Phys Chem B. 2007. PMID: 17228910
-
Adhesion of cultured human endothelial cells onto methacrylate polymers with varying surface wettability and charge.Biomaterials. 1987 Sep;8(5):323-8. doi: 10.1016/0142-9612(87)90001-9. Biomaterials. 1987. PMID: 3676418
-
Keratinocyte Growth Factor-Based Strategies for Wound Re-Epithelialization.Tissue Eng Part B Rev. 2022 Jun;28(3):665-676. doi: 10.1089/ten.TEB.2021.0030. Epub 2021 Oct 18. Tissue Eng Part B Rev. 2022. PMID: 34238035 Review.
Cited by
-
ToF-SIMS and TIRF microscopy investigation on the effects of HEMA copolymer surface chemistry on spatial localization, surface intensity, and release of fluorescently labeled keratinocyte growth factor.Biointerphases. 2019 Sep 23;14(5):051003. doi: 10.1116/1.5119871. Biointerphases. 2019. PMID: 31547664 Free PMC article.
-
Polymer Gels: Classification and Recent Developments in Biomedical Applications.Gels. 2023 Feb 17;9(2):161. doi: 10.3390/gels9020161. Gels. 2023. PMID: 36826331 Free PMC article. Review.
-
Green Hydrogel Synthesis: Emphasis on Proteomics and Polymer Particle-Protein Interaction.Polymers (Basel). 2022 Nov 6;14(21):4755. doi: 10.3390/polym14214755. Polymers (Basel). 2022. PMID: 36365747 Free PMC article. Review.
-
Visualizing Different Crystalline States during the Infrared Imaging of Calcium Phosphates.Vib Spectrosc. 2020 May;108:103045. doi: 10.1016/j.vibspec.2020.103045. Epub 2020 Feb 24. Vib Spectrosc. 2020. PMID: 35360824 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

