Prior to Initiation of Chemotherapy, Can We Predict Breast Tumor Response? Deep Learning Convolutional Neural Networks Approach Using a Breast MRI Tumor Dataset
- PMID: 30361936
- PMCID: PMC6737125
- DOI: 10.1007/s10278-018-0144-1
Prior to Initiation of Chemotherapy, Can We Predict Breast Tumor Response? Deep Learning Convolutional Neural Networks Approach Using a Breast MRI Tumor Dataset
Abstract
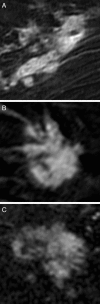
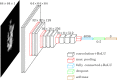
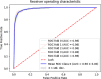
We hypothesize that convolutional neural networks (CNN) can be used to predict neoadjuvant chemotherapy (NAC) response using a breast MRI tumor dataset prior to initiation of chemotherapy. An institutional review board-approved retrospective review of our database from January 2009 to June 2016 identified 141 locally advanced breast cancer patients who (1) underwent breast MRI prior to the initiation of NAC, (2) successfully completed adriamycin/taxane-based NAC, and (3) underwent surgical resection with available final surgical pathology data. Patients were classified into three groups based on their NAC response confirmed on final surgical pathology: complete (group 1), partial (group 2), and no response/progression (group 3). A total of 3107 volumetric slices of 141 tumors were evaluated. Breast tumor was identified on first T1 postcontrast dynamic images and underwent 3D segmentation. CNN consisted of ten convolutional layers, four max-pooling layers, and dropout of 50% after a fully connected layer. Dropout, augmentation, and L2 regularization were implemented to prevent overfitting of data. Non-linear functions were modeled by a rectified linear unit (ReLU). Batch normalization was used between the convolutional and ReLU layers to limit drift of layer activations during training. A three-class neoadjuvant prediction model was evaluated (group 1, group 2, or group 3). The CNN achieved an overall accuracy of 88% in three-class prediction of neoadjuvant treatment response. Three-class prediction discriminating one group from the other two was analyzed. Group 1 had a specificity of 95.1% ± 3.1%, sensitivity of 73.9% ± 4.5%, and accuracy of 87.7% ± 0.6%. Group 2 (partial response) had a specificity of 91.6% ± 1.3%, sensitivity of 82.4% ± 2.7%, and accuracy of 87.7% ± 0.6%. Group 3 (no response/progression) had a specificity of 93.4% ± 2.9%, sensitivity of 76.8% ± 5.7%, and accuracy of 87.8% ± 0.6%. It is feasible for current deep CNN architectures to be trained to predict NAC treatment response using a breast MRI dataset obtained prior to initiation of chemotherapy. Larger dataset will likely improve our prediction model.
Keywords: Breast MRI; Chemotherapy treatment response; Convolutional neural network.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures



Similar articles
-
Predicting Post Neoadjuvant Axillary Response Using a Novel Convolutional Neural Network Algorithm.Ann Surg Oncol. 2018 Oct;25(10):3037-3043. doi: 10.1245/s10434-018-6613-4. Epub 2018 Jul 5. Ann Surg Oncol. 2018. PMID: 29978368
-
Axillary Lymph Node Evaluation Utilizing Convolutional Neural Networks Using MRI Dataset.J Digit Imaging. 2018 Dec;31(6):851-856. doi: 10.1007/s10278-018-0086-7. J Digit Imaging. 2018. PMID: 29696472 Free PMC article.
-
Convolutional Neural Network Using a Breast MRI Tumor Dataset Can Predict Oncotype Dx Recurrence Score.J Magn Reson Imaging. 2019 Feb;49(2):518-524. doi: 10.1002/jmri.26244. Epub 2018 Aug 21. J Magn Reson Imaging. 2019. PMID: 30129697 Free PMC article.
-
3D fully convolutional networks for subcortical segmentation in MRI: A large-scale study.Neuroimage. 2018 Apr 15;170:456-470. doi: 10.1016/j.neuroimage.2017.04.039. Epub 2017 Apr 24. Neuroimage. 2018. PMID: 28450139 Review.
-
Updates on Deep Learning and Glioma: Use of Convolutional Neural Networks to Image Glioma Heterogeneity.Neuroimaging Clin N Am. 2020 Nov;30(4):493-503. doi: 10.1016/j.nic.2020.07.002. Epub 2020 Sep 18. Neuroimaging Clin N Am. 2020. PMID: 33038999 Review.
Cited by
-
The Role of Deep Learning in Advancing Breast Cancer Detection Using Different Imaging Modalities: A Systematic Review.Cancers (Basel). 2022 Oct 29;14(21):5334. doi: 10.3390/cancers14215334. Cancers (Basel). 2022. PMID: 36358753 Free PMC article. Review.
-
A novel CNN algorithm for pathological complete response prediction using an I-SPY TRIAL breast MRI database.Magn Reson Imaging. 2020 Nov;73:148-151. doi: 10.1016/j.mri.2020.08.021. Epub 2020 Sep 2. Magn Reson Imaging. 2020. PMID: 32889091 Free PMC article.
-
Assessing PD-L1 Expression Status Using Radiomic Features from Contrast-Enhanced Breast MRI in Breast Cancer Patients: Initial Results.Cancers (Basel). 2021 Dec 14;13(24):6273. doi: 10.3390/cancers13246273. Cancers (Basel). 2021. PMID: 34944898 Free PMC article.
-
Multimodal Spatiotemporal Deep Learning Framework to Predict Response of Breast Cancer to Neoadjuvant Systemic Therapy.Diagnostics (Basel). 2023 Jul 3;13(13):2251. doi: 10.3390/diagnostics13132251. Diagnostics (Basel). 2023. PMID: 37443648 Free PMC article.
-
Advancing personalized oncology: a systematic review on the integration of artificial intelligence in monitoring neoadjuvant treatment for breast cancer patients.BMC Cancer. 2024 Oct 21;24(1):1300. doi: 10.1186/s12885-024-13049-0. BMC Cancer. 2024. PMID: 39434042 Free PMC article.
References
-
- Rastogi P, Anderson SJ, Bear HD, Geyer CE, Kahlenberg MS, Robidoux A, Margolese RG, Hoehn JL, Vogel VG, Dakhil SR, Tamkus D, King KM, Pajon ER, Wright MJ, Robert J, Paik S, Mamounas EP, Wolmark N. Preoperative chemotherapy: updates of National Surgical Adjuvant Breast and Bowel Project Protocols B-18 and B-27. J Clin Oncol. 2008;26:778–785. doi: 10.1200/JCO.2007.15.0235. - DOI - PubMed
-
- Gianni L, Pienkowski T, Im YH, Tseng LM, Liu MC, Lluch A, Starosławska E, de la Haba-Rodriguez J, Im SA, Pedrini JL, Poirier B, Morandi P, Semiglazov V, Srimuninnimit V, Bianchi GV, Magazzù D, McNally V, Douthwaite H, Ross G, Valagussa P. 5-year analysis of neoadjuvant pertuzumab and trastuzumab in patients with locally advanced, inflammatory, or early-stage HER2-positive breast cancer (NeoSphere): a multicentre, open-label, phase 2 randomised trial. Lancet Oncol. 2016;17:791–800. doi: 10.1016/S1470-2045(16)00163-7. - DOI - PubMed
-
- Cortazar P, Zhang L, Untch M, Mehta K, Costantino JP, Wolmark N, Bonnefoi H, Cameron D, Gianni L, Valagussa P, Swain SM, Prowell T, Loibl S, Wickerham DL, Bogaerts J, Baselga J, Perou C, Blumenthal G, Blohmer J, Mamounas EP, Bergh J, Semiglazov V, Justice R, Eidtmann H, Paik S, Piccart M, Sridhara R, Fasching PA, Slaets L, Tang S, Gerber B, Geyer CE, Jr, Pazdur R, Ditsch N, Rastogi P, Eiermann W, von Minckwitz G. Pathological complete response and long-term clinical benefit in breast cancer: the CTNeoBC pooled analysis. Lancet. 2014;384:164–172. doi: 10.1016/S0140-6736(13)62422-8. - DOI - PubMed
-
- Esserman LJ, Berry DA, DeMichele A, Carey L, Davis SE, Buxton M, Hudis C, Gray JW, Perou C, Yau C, Livasy C, Krontiras H, Montgomery L, Tripathy D, Lehman C, Liu MC, Olopade OI, Rugo HS, Carpenter JT, Dressler L, Chhieng D, Singh B, Mies C, Rabban J, Chen YY, Giri D, van 't Veer L, Hylton N. Pathologic complete response predicts recurrence-free survival more effectively by cancer subset: results from the I-SPY 1 TRIAL--CALGB 150007/150012, ACRIN 6657. J Clin Oncol. 2012;30:3242–3249. doi: 10.1200/JCO.2011.39.2779. - DOI - PMC - PubMed
-
- von Minckwitz G, Untch M, Blohmer JU, Costa SD, Eidtmann H, Fasching PA, Gerber B, Eiermann W, Hilfrich J, Huober J, Jackisch C, Kaufmann M, Konecny GE, Denkert C, Nekljudova V, Mehta K, Loibl S. Definition and impact of pathologic complete response on prognosis after neoadjuvant chemotherapy in various intrinsic breast cancer subtypes. J Clin Oncol. 2012;30:1796–1804. doi: 10.1200/JCO.2011.38.8595. - DOI - PubMed

