IL1-Induced JAK/STAT Signaling Is Antagonized by TGFβ to Shape CAF Heterogeneity in Pancreatic Ductal Adenocarcinoma
- PMID: 30366930
- PMCID: PMC6368881
- DOI: 10.1158/2159-8290.CD-18-0710
IL1-Induced JAK/STAT Signaling Is Antagonized by TGFβ to Shape CAF Heterogeneity in Pancreatic Ductal Adenocarcinoma
Abstract
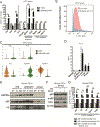
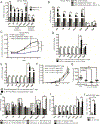
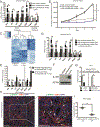
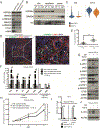
Pancreatic ductal adenocarcinoma (PDAC) is poorly responsive to therapies and histologically contains a paucity of neoplastic cells embedded within a dense desmoplastic stroma. Within the stroma, cancer-associated fibroblasts (CAF) secrete tropic factors and extracellular matrix components, and have been implicated in PDAC progression and chemotherapy resistance. We recently identified two distinct CAF subtypes characterized by either myofibroblastic or inflammatory phenotypes; however, the mechanisms underlying their diversity and their roles in PDAC remain unknown. Here, we use organoid and mouse models to identify TGFβ and IL1 as tumor-secreted ligands that promote CAF heterogeneity. We show that IL1 induces LIF expression and downstream JAK/STAT activation to generate inflammatory CAFs and demonstrate that TGFβ antagonizes this process by downregulating IL1R1 expression and promoting differentiation into myofibroblasts. Our results provide a mechanism through which distinct fibroblast niches are established in the PDAC microenvironment and illuminate strategies to selectively target CAFs that support tumor growth. SIGNIFICANCE: Understanding the mechanisms that determine CAF heterogeneity in PDAC is a prerequisite for the rational development of approaches that selectively target tumor-promoting CAFs. Here, we identify an IL1-induced signaling cascade that leads to JAK/STAT activation and promotes an inflammatory CAF state, suggesting multiple strategies to target these cells in vivo. See related commentary by Ling and Chiao, p. 173. This article is highlighted in the In This Issue feature, p. 151.
©2018 American Association for Cancer Research.
Conflict of interest statement
Figures


Comment in
-
Two Birds with One Stone: Therapeutic Targeting of IL1α Signaling Pathways in Pancreatic Ductal Adenocarcinoma and the Cancer-Associated Fibroblasts.Cancer Discov. 2019 Feb;9(2):173-175. doi: 10.1158/2159-8290.CD-18-1460. Cancer Discov. 2019. PMID: 30737215
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

