Review of methods for intraoperative margin detection for breast conserving surgery
- PMID: 30369108
- PMCID: PMC6210801
- DOI: 10.1117/1.JBO.23.10.100901
Review of methods for intraoperative margin detection for breast conserving surgery
Abstract
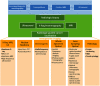
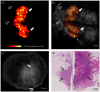
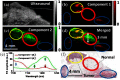
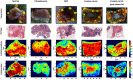
Breast conserving surgery (BCS) is an effective treatment for early-stage cancers as long as the margins of the resected tissue are free of disease according to consensus guidelines for patient management. However, 15% to 35% of patients undergo a second surgery since malignant cells are found close to or at the margins of the original resection specimen. This review highlights imaging approaches being investigated to reduce the rate of positive margins, and they are reviewed with the assumption that a new system would need high sensitivity near 95% and specificity near 85%. The problem appears to be twofold. The first is for complete, fast surface scanning for cellular, structural, and/or molecular features of cancer, in a lumpectomy volume, which is variable in size, but can be large, irregular, and amorphous. A second is for full, volumetric imaging of the specimen at high spatial resolution, to better guide internal radiologic decision-making about the spiculations and duct tracks, which may inform that surfaces are involved. These two demands are not easily solved by a single tool. Optical methods that scan large surfaces quickly are needed with cellular/molecular sensitivity to solve the first problem, but volumetric imaging with high spatial resolution for soft tissues is largely outside of the optical realm and requires x-ray, micro-CT, or magnetic resonance imaging if they can be achieved efficiently. In summary, it appears that a combination of systems into hybrid platforms may be the optimal solution for these two very different problems. This concept must be cost-effective, image specimens within minutes and be coupled to decision-making tools that help a surgeon without adding to the procedure. The potential for optical systems to be involved in this problem is emerging and clinical trials are underway in several of these technologies to see if they could reduce positive margin rates in BCS.
Keywords: breast cancer; breast conserving surgery; imaging; lumpectomy; mammography; optical; spectroscopy.
(2018) COPYRIGHT Society of Photo-Optical Instrumentation Engineers (SPIE).
Figures





Similar articles
-
Micro-computed tomography enables rapid surgical margin assessment during breast conserving surgery (BCS): correlation of whole BCS micro-CT readings to final histopathology.Breast Cancer Res Treat. 2018 Dec;172(3):587-595. doi: 10.1007/s10549-018-4951-3. Epub 2018 Sep 17. Breast Cancer Res Treat. 2018. PMID: 30225621 Free PMC article.
-
Imaging of lumpectomy surface with large field-of-view confocal laser scanning microscopy 'Histolog® scanner' for breast margin assessment in comparison with conventional specimen radiography.Breast. 2023 Apr;68:194-200. doi: 10.1016/j.breast.2023.02.010. Epub 2023 Feb 22. Breast. 2023. PMID: 36842192 Free PMC article.
-
Close/positive margins after breast-conserving therapy: additional resection or no resection?Breast. 2013 Aug;22 Suppl 2:S115-7. doi: 10.1016/j.breast.2013.07.022. Breast. 2013. PMID: 24074771 Review.
-
A comparative study of perioperative techniques to attain negative margins and spare healthy breast tissue in breast conserving surgery.Breast Dis. 2020;39(3-4):127-135. doi: 10.3233/BD-200443. Breast Dis. 2020. PMID: 32831188
-
Biophotonic technologies for assessment of breast tumor surgical margins-A review.J Biophotonics. 2021 Jan;14(1):e202000280. doi: 10.1002/jbio.202000280. Epub 2020 Oct 12. J Biophotonics. 2021. PMID: 32951321 Review.
Cited by
-
Finite Element Modeling of Quantitative Ultrasound Analysis of the Surgical Margin of Breast Tumor.Tomography. 2022 Mar 1;8(2):570-584. doi: 10.3390/tomography8020047. Tomography. 2022. PMID: 35314624 Free PMC article.
-
Real-time diagnosis and visualization of tumor margins in excised breast specimens using fluorescence lifetime imaging and machine learning.Biomed Opt Express. 2020 Feb 14;11(3):1216-1230. doi: 10.1364/BOE.381358. eCollection 2020 Mar 1. Biomed Opt Express. 2020. PMID: 32206404 Free PMC article.
-
Fundamentals and developments in fluorescence-guided cancer surgery.Nat Rev Clin Oncol. 2022 Jan;19(1):9-22. doi: 10.1038/s41571-021-00548-3. Epub 2021 Sep 7. Nat Rev Clin Oncol. 2022. PMID: 34493858 Review.
-
Fluorescence-guided tumor visualization of colorectal cancer using tumor-initiating probe yellow in preclinical models.Sci Rep. 2024 Nov 6;14(1):26946. doi: 10.1038/s41598-024-76312-1. Sci Rep. 2024. PMID: 39505985 Free PMC article.
-
Real-time peri-operative microcalcification detection in superficial breast tissues.Sci Rep. 2025 May 16;15(1):17029. doi: 10.1038/s41598-025-01629-4. Sci Rep. 2025. PMID: 40379809 Free PMC article.
References
-
- Moran M. S., et al. , “Society of Surgical Oncology-American Society for Radiation Oncology Consensus Guideline on margins for breast-conserving surgery with whole-breast irradiation in stages I and II invasive breast cancer,” Ann. Surg. Oncol. 21(3), 704–716 (2014).10.1245/s10434-014-3481-4 - DOI - PubMed
-
- Morrow M., et al. , “Society of Surgical Oncology-American Society for Radiation Oncology-American Society of Clinical Oncology Consensus Guideline on margins for breast-conserving surgery with whole-breast irradiation in ductal carcinoma in situ,” Pract. Radiat. Oncol. 6(5), 287–295 (2016).10.1016/j.prro.2016.06.011 - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

