Prostaglandin E2 Receptor 2 Modulates Macrophage Activity for Cardiac Repair
- PMID: 30371325
- PMCID: PMC6404869
- DOI: 10.1161/JAHA.118.009216
Prostaglandin E2 Receptor 2 Modulates Macrophage Activity for Cardiac Repair
Erratum in
-
Prostaglandin E2 Receptor 2 Modulates Macrophage Activity for Cardiac Repair.J Am Heart Assoc. 2018 Dec 18;7(24):e002261. doi: 10.1161/JAHA.118.002261. J Am Heart Assoc. 2018. PMID: 30545253 Free PMC article. No abstract available.
Abstract
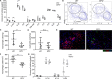
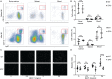
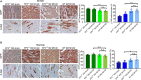
Background Prostaglandin E2 has long been known to be an immune modulator. It is released after tissue injury and plays a role in modulating macrophage activities, which are essential for tissue regeneration. However, the involvement of prostaglandin E2 receptor 2 ( EP 2)-dependent regulation of macrophages in postischemic heart is unclear. This study aims to evaluate the role of EP 2 in damaged heart. Methods and Results The effect of EP 2 in postischemic heart was evaluated using EP 2-deficient transgenic mice. We demonstrated that cardiac function was worse after myocardial injury on loss of EP 2. Furthermore, EP 2 deficiency also altered proinflammatory response and resulted in a defect in macrophage recruitment to the injured myocardium. Transcriptome analysis revealed that the expression of erythroid differentiation regulator 1 ( Erdr1) was significantly induced in EP 2-deficient macrophages. Knocking down Erdr1 expression restored migration ability of EP 2-deficient cells both in vitro and in vivo. By using a genetic fate-mapping approach, we showed that abolishment of EP 2 expression effectively attenuated cell replenishment. Conclusions The EP 2-dependent signaling pathway plays a critical role in regulating macrophage recruitment to the injured myocardium, thereby exerting a function in modulating the inflammatory microenvironment for cardiac repair.
Keywords: EP2; inflammation; ischemia; macrophage; myocardial; therapy.
Figures


References
-
- Sheibanie AF, Tadmori I, Jing H, Vassiliou E, Ganea D. Prostaglandin E2 induces IL‐23 production in bone marrow‐derived dendritic cells. FASEB J. 2004;18:1318–1320. - PubMed
-
- Jing H, Vassiliou E, Ganea D. Prostaglandin E2 inhibits production of the inflammatory chemokines CCL3 and CCL4 in dendritic cells. J Leukoc Biol. 2003;74:868–879. - PubMed
-
- Nemeth K, Leelahavanichkul A, Yuen PS, Mayer B, Parmelee A, Doi K, Robey PG, Leelahavanichkul K, Koller BH, Brown JM, Hu X, Jelinek I, Star RA, Mezey E. Bone marrow stromal cells attenuate sepsis via prostaglandin E(2)‐dependent reprogramming of host macrophages to increase their interleukin‐10 production. Nat Med. 2009;15:42–49. - PMC - PubMed
-
- Murakami M, Naraba H, Tanioka T, Semmyo N, Nakatani Y, Kojima F, Ikeda T, Fueki M, Ueno A, Oh S, Kudo I. Regulation of prostaglandin E2 biosynthesis by inducible membrane‐associated prostaglandin E2 synthase that acts in concert with cyclooxygenase‐2. J Biol Chem. 2000;275:32783–32792. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

