Simultaneous Inhibition of MEK and Hh Signaling Reduces Pancreatic Cancer Metastasis
- PMID: 30373214
- PMCID: PMC6266431
- DOI: 10.3390/cancers10110403
Simultaneous Inhibition of MEK and Hh Signaling Reduces Pancreatic Cancer Metastasis
Abstract
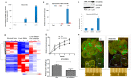
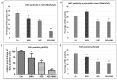
Pancreatic cancer, mostly pancreatic ductal adenocarcinoma (PDAC), is one of the most lethal cancer types, with an estimated 44,330 death in 2018 in the US alone. While targeted therapies and immune checkpoint inhibitors have significantly improved treatment options for patients with lung cancer and renal cell carcinomas, little progress has been made in pancreatic cancer, with a dismal 5-year survival rate currently at ~8%. Upon diagnosis, the majority of pancreatic cancer cases (~80%) are already metastatic. Thus, identifying ways to reduce pancreatic cancer metastasis is an unmet medical need. Furthermore, pancreatic cancer is notorious resistant to chemotherapy. While Kirsten RAt Sarcoma virus oncogene (K-RAS) mutation is the major driver for pancreatic cancer, specific inhibition of RAS signaling has been very challenging, and combination therapy is thought to be promising. In this study, we report that combination of hedgehog (Hh) and Mitogen-activated Protein/Extracellular Signal-regulated Kinase Kinase (MEK) signaling inhibitors reduces pancreatic cancer metastasis in mouse models. In mouse models of pancreatic cancer metastasis using human pancreatic cancer cells, we found that Hh target gene Gli1 is up-regulated during pancreatic cancer metastasis. Specific inhibition of smoothened signaling significantly altered the gene expression profile of the tumor microenvironment but had no significant effects on cancer metastasis. By combining Hh signaling inhibitor BMS833923 with RAS downstream MEK signaling inhibitor AZD6244, we observed reduced number of metastatic nodules in several mouse models for pancreatic cancer metastasis. These two inhibitors also decreased cell proliferation significantly and reduced CD45⁺ cells (particularly Ly6G⁺CD11b⁺ cells). We demonstrated that depleting Ly6G⁺ CD11b⁺ cells is sufficient to reduce cancer cell proliferation and the number of metastatic nodules. In vitro, Ly6G⁺ CD11b⁺ cells can stimulate cancer cell proliferation, and this effect is sensitive to MEK and Hh inhibition. Our studies may help design novel therapeutic strategies to mitigate pancreatic cancer metastasis.
Keywords: Ihh; MEK; hedgehog; metastatic niche; pancreatic cancer.
Conflict of interest statement
The authors declare no competing interests.
Figures




References
-
- Burris H.A., 3rd, Moore M.J., Andersen J., Green M.R., Rothenberg M.L., Modiano M.R., Cripps M.C., Portenoy R.K., Storniolo A.M., Tarassoff P., et al. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: A randomized trial. J. Clin. Oncol. 1997;15:2403–2413. doi: 10.1200/JCO.1997.15.6.2403. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

