Reimagining IDO Pathway Inhibition in Cancer Immunotherapy via Downstream Focus on the Tryptophan-Kynurenine-Aryl Hydrocarbon Axis
- PMID: 30377198
- PMCID: PMC6397695
- DOI: 10.1158/1078-0432.CCR-18-2882
Reimagining IDO Pathway Inhibition in Cancer Immunotherapy via Downstream Focus on the Tryptophan-Kynurenine-Aryl Hydrocarbon Axis
Abstract
Significant progress has been made in cancer immunotherapy with checkpoint inhibitors targeting programmed cell death protein 1 (PD-1)-programmed death-ligand 1 signaling pathways. Tumors from patients showing sustained treatment response predominately demonstrate a T cell-inflamed tumor microenvironment prior to, or early on, treatment. Not all tumors with this phenotype respond, however, and one mediator of immunosuppression in T cell-inflamed tumors is the tryptophan-kynurenine-aryl hydrocarbon receptor (Trp-Kyn-AhR) pathway. Multiple mechanisms of immunosuppression may be mediated by this pathway including depletion of tryptophan, direct immunosuppression of Kyn, and activity of Kyn-bound AhR. Indoleamine 2,3-dioxygenase 1 (IDO1), a principle enzyme in Trp catabolism, is the target of small-molecule inhibitors in clinical development in combination with PD-1 checkpoint inhibitors. Despite promising results in early-phase clinical trials in a range of tumor types, a phase III study of the IDO1-selective inhibitor epacadostat in combination with pembrolizumab showed no difference between the epacadostat-treated group versus placebo in patients with metastatic melanoma. This has led to a diminution of interest in IDO1 inhibitors; however, other approaches to inhibit this pathway continue to be considered. Novel Trp-Kyn-AhR pathway inhibitors, such as Kyn-degrading enzymes, direct AhR antagonists, and tryptophan mimetics are advancing in early-stage or preclinical development. Despite uncertainty surrounding IDO1 inhibition, ample preclinical evidence supports continued development of Trp-Kyn-AhR pathway inhibitors to augment immune-checkpoint and other cancer therapies.
©2018 American Association for Cancer Research.
Conflict of interest statement
Conflicts of Interest: JJL Consultancies to: 7 Hills, Actym, Amgen, Array, AstraZeneca, Bristol-Myers Squibb, Castle, CheckMate, Compugen, EMD Serono, Gilead, Ideaya, Janssen, Merck, NewLink, Novartis, Palleon, RefleXion, Syndax, Tempest, WntRx with research support from AbbVie, Array, Boston Biomedical, Bristol-Myers Squibb, Celldex, CheckMate, Corvus, Delcath, Five Prime, Genentech, Immunocore, Incyte, Leap, MedImmune, Macrogenics, Novartis, Pharmacyclics, Palleon, Merck, Tesaro, Xencor
Figures


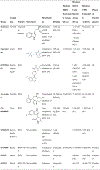

References
-
- Rosenberg SA, Sherry RM, Morton KE, Scharfman WJ, Yang JC, Topalian SL, et al. Tumor progression can occur despite the induction of very high levels of self/tumor antigen-specific CD8+ T cells in patients with melanoma. J Immunol 2005;175(9):6169–76. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

