Recent progress of porcine milk components and mammary gland function
- PMID: 30377527
- PMCID: PMC6196465
- DOI: 10.1186/s40104-018-0291-8
Recent progress of porcine milk components and mammary gland function
Abstract
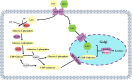
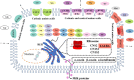
As the only nutritional source for newborn piglets, porcine colostrum and milk contain critical nutritional and immunological components including carbohydrates, lipids, and proteins (immunoglobulins). However, porcine milk composition is more complex than these three components. Recently, scientists identified additional and novel components of sow colostrum and milk, including exosomes, oligosaccharides, and bacteria, which possibly act as biological signals and modulate the intestinal environment and immune status in piglets and later in life. Evaluation of these nutritional and non-nutritional components in porcine milk will help better understand the nutritional and biological function of porcine colostrum and milk. Furthermore, some important functions of the porcine mammary gland have been reported in recent published literature. These preliminary studies hypothesized how glucose, amino acids, and fatty acids are transported from maternal blood to the porcine mammary gland for milk synthesis. Therefore, we summarized recent reports on sow milk composition and porcine mammary gland function in this review, with particular emphasis on macronutrient transfer and synthesis mechanisms, which might offer a possible approach for regulation of milk synthesis in the future.
Keywords: Bioactive components; Fat; Lactose; Mammary gland; Porcine milk; Protein.
Conflict of interest statement
Not applicable.The authors declare that they have no competing interests.
Figures


Similar articles
-
Review: nutritional and endocrine control of colostrogenesis in swine.Animal. 2019 Jul;13(S1):s26-s34. doi: 10.1017/S1751731118003555. Animal. 2019. PMID: 31280746 Review.
-
Composition and physiological functions of the porcine colostrum.Anim Sci J. 2021 Jan-Dec;92(1):e13618. doi: 10.1111/asj.13618. Anim Sci J. 2021. PMID: 34409709 Free PMC article. Review.
-
Characterization and comparative analysis of transcriptional profiles of porcine colostrum and mature milk at different parities.BMC Genom Data. 2021 Aug 10;22(1):25. doi: 10.1186/s12863-021-00980-5. BMC Genom Data. 2021. PMID: 34376140 Free PMC article.
-
Valine increases milk fat synthesis in mammary gland of gilts through stimulating AKT/MTOR/SREBP1 pathway†.Biol Reprod. 2019 Jul 1;101(1):126-137. doi: 10.1093/biolre/ioz065. Biol Reprod. 2019. PMID: 30985894
-
Neonatal piglet survival: impact of sow nutrition around parturition on fetal glycogen deposition and production and composition of colostrum and transient milk.Animal. 2014 Jul;8(7):1021-30. doi: 10.1017/S1751731114000950. Epub 2014 Apr 25. Animal. 2014. PMID: 24762853 Review.
Cited by
-
Euglena gracilis β-Glucans (1,3): Enriching Colostrum of Sow for Enhanced Piglet Immunity.Animals (Basel). 2023 Nov 12;13(22):3490. doi: 10.3390/ani13223490. Animals (Basel). 2023. PMID: 38003108 Free PMC article.
-
Impacts of Maternal Nutrition on Sow Performance and Potential Positive Effects on Piglet Performance.Animals (Basel). 2024 Jun 23;14(13):1858. doi: 10.3390/ani14131858. Animals (Basel). 2024. PMID: 38997970 Free PMC article. Review.
-
Clostridium butyricum Probiotic Feed Additive: Modulation of Sow Milk Metabolomics and Mitigation of Pre-Weaning Piglet Diarrhea.Animals (Basel). 2024 Jul 18;14(14):2098. doi: 10.3390/ani14142098. Animals (Basel). 2024. PMID: 39061560 Free PMC article.
-
Feeding with 4,4'-diaponeurosporene-producing Bacillus subtilis enhances the lactogenic immunity of sow.BMC Vet Res. 2023 Dec 19;19(1):280. doi: 10.1186/s12917-023-03846-3. BMC Vet Res. 2023. PMID: 38115003 Free PMC article.
-
A Pair of Fluorescent Probes Enabling Precise Diagnosis of Liver Cancer by Complementary Imaging.ACS Cent Sci. 2024 Dec 16;11(1):76-83. doi: 10.1021/acscentsci.4c01822. eCollection 2025 Jan 22. ACS Cent Sci. 2024. PMID: 39866701 Free PMC article.
References
-
- Le Dividich J, Rooke JA, Herpin P. Nutritional and immunological importance of colostrum for the new-born pig. J Agr Sci. 2005;143:469–485. doi: 10.1017/S0021859605005642. - DOI
-
- Mei J, Zhang YQ, Wang T, Sangild PT, Xu RJ. Oral ingestion of colostrum alters intestinal transforming growth factor-beta receptor intensity in newborn pigs. Livest Sci. 2006;105:214–222. doi: 10.1016/j.livsci.2006.06.017. - DOI
-
- Quesnel H, Farmer C, Devillers N. Colostrum intake: influence on piglet performance and factors of variation. Livest Sci. 2012;146:105–114. doi: 10.1016/j.livsci.2012.03.010. - DOI
Publication types
LinkOut - more resources
Full Text Sources
Research Materials

