Effects of glucagon-like peptide 1 analogs on alcohol intake in alcohol-preferring vervet monkeys
- PMID: 30382353
- PMCID: PMC6428196
- DOI: 10.1007/s00213-018-5089-z
Effects of glucagon-like peptide 1 analogs on alcohol intake in alcohol-preferring vervet monkeys
Erratum in
-
Correction to: Effects of glucagon-like peptide 1 analogs on alcohol intake in alcohol-preferring vervet monkeys.Psychopharmacology (Berl). 2020 Jan;237(1):287. doi: 10.1007/s00213-019-05374-1. Psychopharmacology (Berl). 2020. PMID: 31768561
Abstract
Background: Preclinical studies in rodents have demonstrated inhibitory effects of glucagon-like peptide-1 (GLP-1) receptor stimulation on alcohol consumption. The effects of GLP-1 receptor stimulation on alcohol intake in primates have not been investigated.
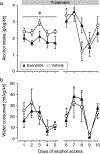
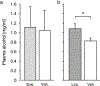
Methods: We performed placebo-controlled studies on the effects of the GLP-1 receptor agonists exenatide and liraglutide on alcohol consumption in alcohol-preferring male African vervet monkeys. Monkeys selected for voluntary alcohol drinking were observed for at least 10 days of baseline drinking and allocated to drug or vehicle (n = 11-12 per group) balanced with respect to alcohol intake. Monkeys had access to alcohol 4 h/day. In a first study, monkeys were treated with exenatide 0.04 mg/kg or vehicle once weekly for 5 weeks to obtain steady-state plasma levels. In a second study, monkeys were treated daily with liraglutide (increasing dosing, 10 to 50 μg/kg/day) or vehicle over 2 weeks. In both studies, access to alcohol was suspended during drug up-titration. Then, alcohol was again made available 4 h/day and treatment was continued for 2 weeks, during which alcohol intake was recorded. Observation of alcohol intake was continued for a week of drug washout.
Results: Liraglutide and to a lesser extent exenatide significantly reduced alcohol consumption without causing any signs of emesis and with no effect on water intake as compared to vehicle.
Conclusions: The present study demonstrates for the first time that GLP-1 receptor agonists can reduce voluntary alcohol drinking in non-human primates. The data substantiate the potential usefulness of GLP-1 receptor agonists in the treatment of alcohol use disorder.
Keywords: Alcohol use disorder; Exendin-4; GLP-1; Liraglutide; Non-human primate; Pharmacotherapy.
Conflict of interest statement
The authors declare no biomedical financial interests or potential conflicts of interest in relation to the work described.
Figures



References
-
- (WHO) World Health Organization (2014) Global status report on alcohol and health 2014. WHO Library Cataloguing-in-Publication Data 2014; ed.
-
- Antonsen KK, Kruse MK, Brunchmann AS, le Dous N, Jensen ME, Miskowiak KW, Fisher PM, Thomsen GK, Rindom H, Fahmy TP, Vollstädt-Klein S, Benveniste H, Volkow ND, Becker U, Ekstrøm C, Knudsen GM, Vilsbøll T, Fink-Jensen A. (2018) Does glucagon-like peptide-1 (GLP-1) receptor stimulation reduce alcohol intake in patients with alcohol dependence? Study protocol of a randomized, double-blinded, placebo-controlled clinical trial. BMJ Open 8:e019562. doi:10.1136/bmjopen-2017-019562. - DOI - PMC - PubMed
-
- Buse JB, Garber A, Rosenstock J, Schmidt WE, Brett JH, Videbaek N, Holst J, Nauck M (2011) Liraglutide treatment is associated with a low frequency and magnitude of antibody formation with no apparent impact on glycemic response or increased frequency of adverse events: results from the Liraglutide Effect and Action in Diabetes (LEAD) trials. J Clin Endocrinol Metab 96: 1695–702. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

