Genetic Strategies for HIV Treatment and Prevention
- PMID: 30388625
- PMCID: PMC6205348
- DOI: 10.1016/j.omtn.2018.09.018
Genetic Strategies for HIV Treatment and Prevention
Abstract
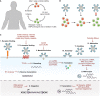
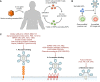
Conventional HIV gene therapy approaches are based on engineering HIV target cells that are non-permissive to viral replication. However, expansion of gene-modified HIV target cells has been limited in patients. Alternative genetic strategies focus on generating gene-modified producer cells that secrete antiviral proteins (AVPs). The secreted AVPs interfere with HIV entry, and, therefore, they extend the protection against infection to unmodified HIV target cells. Since any cell type can potentially secrete AVPs, hematopoietic and non-hematopoietic cell lineages can function as producer cells. Secretion of AVPs from non-hematopoietic cells opens the possibility of using a genetic approach for HIV prevention. Another strategy aims at modifying cytotoxic T cells to selectively target and eliminate infected cells. This review provides an overview of the different genetic approaches for HIV treatment and prevention.
Keywords: HIV; antiviral proteins; genetic therapies.
Copyright © 2018 The Authors. Published by Elsevier Inc. All rights reserved.
Figures
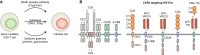
Similar articles
-
A novel gene therapy strategy using secreted multifunctional anti-HIV proteins to confer protection to gene-modified and unmodified target cells.Gene Ther. 2014 Feb;21(2):175-87. doi: 10.1038/gt.2013.70. Epub 2013 Dec 5. Gene Ther. 2014. PMID: 24305417
-
HIV Gene Therapy Strategies and Safety: What do we know from the Recent Publications?AIDS Rev. 2020 Oct 26;23(3):195-202. doi: 10.24875/AIDSRev.20000008. AIDS Rev. 2020. PMID: 33105475
-
Control of HIV Infection In Vivo Using Gene Therapy with a Secreted Entry Inhibitor.Mol Ther Nucleic Acids. 2017 Dec 15;9:132-144. doi: 10.1016/j.omtn.2017.08.017. Epub 2017 Sep 21. Mol Ther Nucleic Acids. 2017. PMID: 29246292 Free PMC article.
-
Current developments and future prospects for HIV gene therapy using interfering RNA-based strategies.Front Biosci. 2000 May 1;5:D527-55. doi: 10.2741/lamothe. Front Biosci. 2000. PMID: 10799355 Review.
-
C peptides as entry inhibitors for gene therapy.Adv Exp Med Biol. 2015;848:191-209. doi: 10.1007/978-1-4939-2432-5_10. Adv Exp Med Biol. 2015. PMID: 25757622 Review.
Cited by
-
HIV-1/HBV Coinfection Accurate Multitarget Prediction Using a Graph Neural Network-Based Ensemble Predicting Model.Int J Mol Sci. 2023 Apr 12;24(8):7139. doi: 10.3390/ijms24087139. Int J Mol Sci. 2023. PMID: 37108305 Free PMC article.
-
Hepatitis B and HIV-1 2019 IAS Cure Forum: lessons and benefits from interdisciplinary research.J Virus Erad. 2019 Nov 4;5(4):234-244. doi: 10.1016/S2055-6640(20)30027-3. J Virus Erad. 2019. PMID: 31754448 Free PMC article. No abstract available.
-
Kinases: Understanding Their Role in HIV Infection.World J AIDS. 2019 Sep;9(3):142-160. doi: 10.4236/wja.2019.93011. Epub 2019 Sep 9. World J AIDS. 2019. PMID: 32257606 Free PMC article.
-
HIV Infection Predisposes to Increased Chances of HBV Infection: Current Understanding of the Mechanisms Favoring HBV Infection at Each Clinical Stage of HIV Infection.Front Immunol. 2022 Apr 1;13:853346. doi: 10.3389/fimmu.2022.853346. eCollection 2022. Front Immunol. 2022. PMID: 35432307 Free PMC article. Review.
-
A Membrane-Anchored Short-Peptide Fusion Inhibitor Fully Protects Target Cells from Infections of Human Immunodeficiency Virus Type 1 (HIV-1), HIV-2, and Simian Immunodeficiency Virus.J Virol. 2019 Oct 29;93(22):e01177-19. doi: 10.1128/JVI.01177-19. Print 2019 Nov 15. J Virol. 2019. PMID: 31462566 Free PMC article.
References
-
- UNAIDS. (2016). Global AIDS update. Joint United Nations Programme on HIV/AIDS. http://www.unaids.org/sites/default/files/media_asset/global-AIDS-update.... - PubMed
-
- Hütter G., Nowak D., Mossner M., Ganepola S., Müssig A., Allers K., Schneider T., Hofmann J., Kücherer C., Blau O. Long-term control of HIV by CCR5 Delta32/Delta32 stem-cell transplantation. N. Engl. J. Med. 2009;360:692–698. - PubMed
-
- Allers K., Hütter G., Hofmann J., Loddenkemper C., Rieger K., Thiel E., Schneider T. Evidence for the cure of HIV infection by CCR5Δ32/Δ32 stem cell transplantation. Blood. 2011;117:2791–2799. - PubMed
-
- Schacker T., Collier A.C., Coombs R., Unadkat J.D., Fox I., Alam J., Wang J.P., Eggert E., Corey L. Phase I study of high-dose, intravenous rsCD4 in subjects with advanced HIV-1 infection. J. Acquir. Immune Defic. Syndr. Hum. Retrovirol. 1995;9:145–152. - PubMed
Publication types
LinkOut - more resources
Full Text Sources

