CD7 CAR T Cells for the Therapy of Acute Myeloid Leukemia
- PMID: 30391141
- PMCID: PMC6318703
- DOI: 10.1016/j.ymthe.2018.10.001
CD7 CAR T Cells for the Therapy of Acute Myeloid Leukemia
Abstract
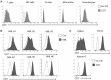
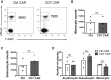
Chimeric antigen receptor (CAR) T cell therapy for the treatment of acute myeloid leukemia (AML) has the risk of toxicity to normal myeloid cells. CD7 is expressed by the leukemic blasts and malignant progenitor cells of approximately 30% of AML patients but is absent on normal myeloid and erythroid cells. Since CD7 expression by malignant blasts is also linked with chemoresistance and poor outcomes, targeting this antigen may be beneficial for this subset of AML patients. Here, we show that expression of a CD7-directed CAR in CD7 gene-edited (CD7KO) T cells effectively eliminates CD7+ AML cell lines, primary CD7+ AML, and colony-forming cells but spares myeloid and erythroid progenitor cells and their progeny. In a xenograft model, CD7 CAR T cells protect mice against systemic leukemia, prolonging survival. Our results support the feasibility of using CD7KO CD7 CAR T cells for the non-myeloablative treatment of CD7+ AML.
Keywords: CAR T cells; CD7; acute myeloid leukemia; chimeric antigen receptors; mouse model.
Copyright © 2018 The American Society of Gene and Cell Therapy. Published by Elsevier Inc. All rights reserved.
Figures



References
-
- Mardiros A., Dos Santos C., McDonald T., Brown C.E., Wang X., Budde L.E., Hoffman L., Aguilar B., Chang W.C., Bretzlaff W. T cells expressing CD123-specific chimeric antigen receptors exhibit specific cytolytic effector functions and antitumor effects against human acute myeloid leukemia. Blood. 2013;122:3138–3148. - PMC - PubMed
-
- Westwood J.A., Smyth M.J., Teng M.W.L., Moeller M., Trapani J.A., Scott A.M., Smyth F.E., Cartwright G.A., Power B.E., Hönemann D. Adoptive transfer of T cells modified with a humanized chimeric receptor gene inhibits growth of Lewis-Y-expressing tumors in mice. Proc. Natl. Acad. Sci. USA. 2005;102:19051–19056. - PMC - PubMed
-
- Tashiro H., Sauer T., Shum T., Parikh K., Mamonkin M., Omer B., Rouce R.H., Lulla P., Rooney C.M., Gottschalk S., Brenner M.K. Treatment of Acute Myeloid Leukemia with T Cells Expressing Chimeric Antigen Receptors Directed to C-type Lectin-like Molecule 1. Mol. Ther. 2017;25:2202–2213. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

