Does Age Interfere With Gadolinium Toxicity and Presence in Brain and Bone Tissues?: A Comparative Gadoterate Versus Gadodiamide Study in Juvenile and Adult Rats
- PMID: 30394964
- PMCID: PMC6310471
- DOI: 10.1097/RLI.0000000000000517
Does Age Interfere With Gadolinium Toxicity and Presence in Brain and Bone Tissues?: A Comparative Gadoterate Versus Gadodiamide Study in Juvenile and Adult Rats
Abstract
Objectives: The main objective of the study was to assess the effect of age on target tissue total gadolinium (Gd) retention after repeated administration of gadodiamide (linear) or gadoterate (macrocyclic) Gd-based contrast agent (GBCA) in rats. The secondary objective was to assess the potential developmental and long-term consequences of GBCA administration during neonatal and juvenile periods.
Materials and methods: A total of 20 equivalent human clinical doses (cumulated dose, 12 mmol Gd/kg) of either gadoterate or gadodiamide were administered concurrently by the intravenous route to healthy adult and juvenile rats. Saline was administered to juvenile rats forming the control group. In juvenile rats, the doses were administered from postnatal day 12, that is, once the blood-brain barrier is functional as in humans after birth. The tests were conducted on 5 juvenile rats per sex and per group and on 3 adult animals per sex and per group. T1-weighted magnetic resonance imaging of the cerebellum was performed at 4.7 T during both the treatment and treatment-free periods. Behavioral tests were performed in juvenile rats. Rats were euthanatized at 11 to 12 weeks (ie, approximately 3 months) after the last administration. Total Gd concentrations were measured in plasma, skin, bone, and brain by inductively coupled plasma mass spectrometry. Cerebellum samples from the juvenile rats were characterized by histopathological examination (including immunohistochemistry for glial fibrillary acidic protein or GFAP, and CD68). Lipofuscin pigments were also studied by fluorescence microscopy. All tests were performed blindly on randomized animals.
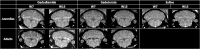
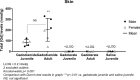
Results: Transient skin lesions were observed in juvenile rats (5/5 females and 2/4 males) and not in adult rats having received gadodiamide. Persisting (up to completion of the study) T1 hyperintensity in the deep cerebellar nuclei (DCNs) was observed only in gadodiamide-treated rats. Quantitatively, a slightly higher progressive increase in the DCN/brain stem ratio was observed in adult rats compared with juvenile rats, whereas no difference was noted visually. In all tissues, total Gd concentrations were higher (10- to 30-fold higher) in the gadodiamide-treated groups than in the gadoterate groups. No age-related differences were observed except in bone marrow where total Gd concentrations in gadodiamide-treated juvenile rats were higher than those measured in adults and similar to those measured in cortical bone tissue. No significant treatment-related effects were observed in histopathological findings or in development, behavior, and biochemistry parameters. However, in the elevated plus maze test, a trend toward an anxiogenic effect was observed in the gadodiamide group compared with other groups (nonsignificant). Moreover, in the balance beam test, a high number of trials were excluded in the gadodiamide group because rats (mainly males) did not completely cross the beam, which may also reflect an anxiogenic effect.
Conclusions: No T1 hyperintensity was observed in the DCN after administration of the macrocyclic GBCA gadoterate regardless of age as opposed to administration of the linear GBCA gadodiamide. Repeated administration of gadodiamide in neonatal and juvenile rats resulted in similar total Gd retention in the skin, brain, and bone to that in adult rats with sex having no effect, whereas Gd distribution in bone marrow was influenced by age. Further studies are required to assess the form of the retained Gd and to investigate the potential risks associated with Gd retention in bone marrow in juvenile animals treated with gadodiamide. Regardless of age, total Gd concentration in the brain and bone was 10- to 30-fold higher after administration of gadodiamide compared with gadoterate.
Conflict of interest statement
Conflicts of interest and sources of funding: All authors (except F.B.) were Guebert employees at the time of the study. This study was funded by Guebert through the Atlantic Bone Screen funding within the common research project.
Figures







Comment in
-
The Critical Need for Pediatric and Juvenile Animal Research Addressing Gadolinium Retention in the Developing Body.Invest Radiol. 2019 Feb;54(2):72-75. doi: 10.1097/RLI.0000000000000516. Invest Radiol. 2019. PMID: 30273280 No abstract available.
References
-
- Girardi M, Kay J, Elston DM, et al. Nephrogenic systemic fibrosis: clinicopathological definition and workup recommendations. J Am Acad Dermatol. 2011;65:1095–1196. - PubMed
-
- Grobner T. Gadolinium—a specific trigger for the development of nephrogenic fibrosing dermopathy and nephrogenic systemic fibrosis? Nephrol Dial Transplant. 2006;21:1104–1108. - PubMed
-
- Thomsen HS, Morcos SK, Almén T, et al. Nephrogenic systemic fibrosis and gadolinium-based contrast media: updated ESUR contrast medium safety committee guidelines. Eur Radiol. 2013;23:307–318. - PubMed
-
- American College of Radiology. ACR manual on contrast media: Version 10.3. Am Coll Radiol. 2018.
-
- Yang L, Krefting I, Gorovets A, et al. Nephrogenic systemic fibrosis and class labeling of gadolinium-based contrast agents by the food and drug administration. Radiology. 2012;265:248–253. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

