Genetic predisposition for inflammation exacerbates effects of striatal iron content on cognitive switching ability in healthy aging
- PMID: 30395929
- PMCID: PMC6301022
- DOI: 10.1016/j.neuroimage.2018.10.064
Genetic predisposition for inflammation exacerbates effects of striatal iron content on cognitive switching ability in healthy aging
Abstract
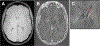
Non-heme iron homeostasis interacts with inflammation bidirectionally, and both contribute to age-related decline in brain structure and function via oxidative stress. Thus, individuals with genetic predisposition for inflammation may be at greater risk for brain iron accumulation during aging and more vulnerable to cognitive decline. We examine this hypothesis in a lifespan sample of healthy adults (N = 183, age 20-94 years) who underwent R2*-weighted magnetic resonance imaging to estimate regional iron content and genotyping of interleukin-1beta (IL-1β), a pro-inflammatory cytokine for which the T allelle of the single nucleotide polymorphism increases risk for chronic neuroinflammation. Older age was associated with greater striatal iron content that in turn accounted for poorer cognitive switching performance. Heterozygote IL-1β T-carriers demonstrated poorer switching performance in relation to striatal iron content as compared to IL-1β C/C counterparts, despite the two groups being of similar age. With increasing genetic inflammation risk, homozygote IL-1β T/T carriers had lesser age-related variance in striatal iron content as compared to the other groups but showed a similar association of greater striatal iron content predicting poorer cognitive switching. Non-heme iron and inflammation, although necessary for normal neuronal function, both promote oxidative stress that when accumulated in excess, drives a complex mechanism of neural and cognitive decline in aging.
Keywords: Aging; Inflammation; Iron; R2* imaging; Structural equation modeling; Task switching.
Copyright © 2018 The Authors. Published by Elsevier Inc. All rights reserved.
Figures
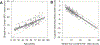

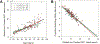
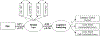
Similar articles
-
Striatal iron content is linked to reduced fronto-striatal brain function under working memory load.Neuroimage. 2020 Apr 15;210:116544. doi: 10.1016/j.neuroimage.2020.116544. Epub 2020 Jan 20. Neuroimage. 2020. PMID: 31972284 Free PMC article.
-
Striatal iron content predicts its shrinkage and changes in verbal working memory after two years in healthy adults.J Neurosci. 2015 Apr 29;35(17):6731-43. doi: 10.1523/JNEUROSCI.4717-14.2015. J Neurosci. 2015. PMID: 25926451 Free PMC article.
-
Higher Striatal Iron Concentration is Linked to Frontostriatal Underactivation and Poorer Memory in Normal Aging.Cereb Cortex. 2017 Jun 1;27(6):3427-3436. doi: 10.1093/cercor/bhx045. Cereb Cortex. 2017. PMID: 28334149
-
Appraising the Role of Iron in Brain Aging and Cognition: Promises and Limitations of MRI Methods.Neuropsychol Rev. 2015 Sep;25(3):272-87. doi: 10.1007/s11065-015-9292-y. Epub 2015 Aug 7. Neuropsychol Rev. 2015. PMID: 26248580 Free PMC article. Review.
-
The impact of brain iron accumulation on cognition: A systematic review.PLoS One. 2020 Oct 15;15(10):e0240697. doi: 10.1371/journal.pone.0240697. eCollection 2020. PLoS One. 2020. PMID: 33057378 Free PMC article.
Cited by
-
Striatal iron content is linked to reduced fronto-striatal brain function under working memory load.Neuroimage. 2020 Apr 15;210:116544. doi: 10.1016/j.neuroimage.2020.116544. Epub 2020 Jan 20. Neuroimage. 2020. PMID: 31972284 Free PMC article.
-
R2* and quantitative susceptibility mapping in deep gray matter of 498 healthy controls from 5 to 90 years.Hum Brain Mapp. 2021 Oct 1;42(14):4597-4610. doi: 10.1002/hbm.25569. Epub 2021 Jun 29. Hum Brain Mapp. 2021. PMID: 34184808 Free PMC article.
-
Nigral Iron Deposition Influences Disease Severity by Modulating the Effect of Parkinson's Disease on Brain Networks.J Parkinsons Dis. 2022;12(8):2479-2492. doi: 10.3233/JPD-223372. J Parkinsons Dis. 2022. PMID: 36336939 Free PMC article.
-
Inflaming the Brain with Iron.Antioxidants (Basel). 2021 Jan 6;10(1):61. doi: 10.3390/antiox10010061. Antioxidants (Basel). 2021. PMID: 33419006 Free PMC article. Review.
-
Low-dose proton induced genetic alteration in cingulate cortex and declined its relevant cognitive function in behaviors.Front Behav Neurosci. 2025 Mar 10;19:1514579. doi: 10.3389/fnbeh.2025.1514579. eCollection 2025. Front Behav Neurosci. 2025. PMID: 40130225 Free PMC article.
References
-
- Adamo DE, Daugherty AM, Raz N, 2014. Grasp force matching and brain iron content estimated in vivo in older women. Brain Imaging Behav. 8, 579–587. https://doi.org/10.1007/s11682-013-9284-6 - DOI - PubMed
-
- Akiyama H, Barger S, Barnum S, Bradt B, Bauer J, Cole GM, Cooper NR, Eikelenboom P, Emmerling M, Fiebich BL, Finch CE, Frautschy S, Griffin WST, Hampel H, Hull M, Landreth G, Lue L, Mrak R, Mackenzie IR, McGeer PL, O’Banion MK, Pachter J, Pasinetti G, Plata-Salaman C, Rogers J, Rydel R, Shen Y, Streit W, Strohmeyer R, Tooyoma I, Van Muiswinkel FL, Veerhuis R, Walker D, Webster S, Wegrzyniak B, Wenk G, Wyss-Coray T, 2000. Inflammation and Alzheimer’s disease. Neurobiol. Aging 21, 383–421. - PMC - PubMed
-
- Andrews NC, 2000. Iron homeostasis: Insights from genetics and animal models. Nat. Rev. Genet. 1, 208–217. - PubMed
-
- Banks WA, 2005. Blood-Brain Barrier Transport of Cytokines: A Mechanism for Neuropathology [WWW Document]. Curr. Pharm. Des. URL http://www.eurekaselect.com/60690/article (accessed 6.25.18). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

