Vascular permeability in the brain is a late pathogenic event during Rift Valley fever virus encephalitis in rats
- PMID: 30396029
- PMCID: PMC6286220
- DOI: 10.1016/j.virol.2018.10.021
Vascular permeability in the brain is a late pathogenic event during Rift Valley fever virus encephalitis in rats
Abstract
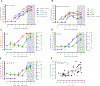
Rift Valley fever virus (RVFV) is a zoonotic disease of livestock that causes several clinical outcomes in people including febrile disease, hemorrhagic fever, and/or encephalitis. After aerosol infection with RVFV, Lewis rats develop lethal encephalitic disease, and we use this as a model for studying disease mechanisms of RVFV infection in the brain. Permeability of the brain vasculature in relation to virus invasion and replication is not known. Here, we found that vascular permeability in the brain occurred late in the course of infection and corresponded temporally to expression of matrix metalloproteinase-9 (MMP-9). Virus replication was ongoing within the central nervous system for several days prior to detectable vascular leakage. Based on this study, vascular permeability was not required for entry of RVFV into the brain of rats. Prevention of vascular leakage late in infection may be an important component for prevention of lethal neurological disease in the rat model.
Keywords: Aerosol infection; Blood brain barrier; Rift valley fever; Viral encephalitis.
Copyright © 2018 Elsevier Inc. All rights reserved.
Figures
References
-
- Al-Hazmi M, Ayoola EA, Abdurahman M, Banzal S, Ashraf J, El-Bushra A, Hazmi A, Abdullah M, Abbo H, Elamin A, Al-Sammani el T, Gadour M, Menon C, Hamza M, Rahim I, Hafez M, Jambavalikar M, Arishi H, Aqeel A, 2003. Epidemic Rift Valley fever in Saudi Arabia: a clinical study of severe illness in humans. Clin Infect Dis 36, 245–252. - PubMed
-
- Bird BH, Reynes JM, Nichol ST, 2012. Rift Valley Fever, in: Magill AJ, Strickland GT, Maguire JH, Ryan ET, Solomon T (Eds.), Hunter’s Tropical Medicine and Emerging Infectious Disease, 9th ed. Elsevier Health Sciences, pp. 340–343.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

