Muscle strength deficiency and mitochondrial dysfunction in a muscular dystrophy model of Caenorhabditis elegans and its functional response to drugs
- PMID: 30396907
- PMCID: PMC6307913
- DOI: 10.1242/dmm.036137
Muscle strength deficiency and mitochondrial dysfunction in a muscular dystrophy model of Caenorhabditis elegans and its functional response to drugs
Abstract
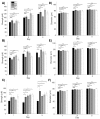
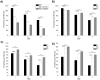
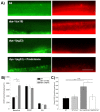
Muscle strength is a key clinical parameter used to monitor the progression of human muscular dystrophies, including Duchenne and Becker muscular dystrophies. Although Caenorhabditis elegans is an established genetic model for studying the mechanisms and treatments of muscular dystrophies, analogous strength-based measurements in this disease model are lacking. Here, we describe the first demonstration of the direct measurement of muscular strength in dystrophin-deficient C. elegans mutants using a micropillar-based force measurement system called NemaFlex. We show that dys-1(eg33) mutants, but not dys-1(cx18) mutants, are significantly weaker than their wild-type counterparts in early adulthood, cannot thrash in liquid at wild-type rates, display mitochondrial network fragmentation in the body wall muscles, and have an abnormally high baseline mitochondrial respiration. Furthermore, treatment with prednisone, the standard treatment for muscular dystrophy in humans, and melatonin both improve muscular strength, thrashing rate and mitochondrial network integrity in dys-1(eg33), and prednisone treatment also returns baseline respiration to normal levels. Thus, our results demonstrate that the dys-1(eg33) strain is more clinically relevant than dys-1(cx18) for muscular dystrophy studies in C. elegans This finding, in combination with the novel NemaFlex platform, can be used as an efficient workflow for identifying candidate compounds that can improve strength in the C. elegans muscular dystrophy model. Our study also lays the foundation for further probing of the mechanism of muscle function loss in dystrophin-deficient C. elegans, leading to knowledge translatable to human muscular dystrophy.This article has an associated First Person interview with the first author of the paper.
Keywords: C. elegans; Melatonin; Muscle strength; Muscular dystrophy; Prednisone.
© 2018. Published by The Company of Biologists Ltd.
Conflict of interest statement
Competing interestsThe authors declare no competing or financial interests.
Figures
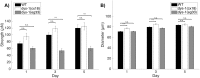



References
-
- Brouilly N., Lecroisey C., Martin E., Pierson L., Mariol M.-C., Qadota H., Labouesse M., Streichenberger N., Mounier N. and Gieseler K. (2015). Ultra-structural time-course study in the C. elegans model for Duchenne muscular dystrophy highlights a crucial role for sarcomere-anchoring structures and sarcolemma integrity in the earliest steps of the muscle degeneration process. Hum. Mol. Genet. 24, 6428-6445. 10.1093/hmg/ddv353 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

