Pharmacodynamics of Minocycline against Acinetobacter baumannii in a Rat Pneumonia Model
- PMID: 30397059
- PMCID: PMC6355566
- DOI: 10.1128/AAC.01671-18
Pharmacodynamics of Minocycline against Acinetobacter baumannii in a Rat Pneumonia Model
Abstract
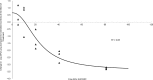
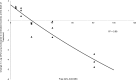
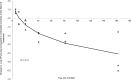
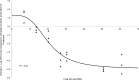
Minocycline is currently approved in the United States for the treatment of infections caused by susceptible isolates of Acinetobacter spp. The objective of these studies was to determine the minocycline exposures associated with an antibacterial effect against Acinetobacter baumannii in a rat pneumonia model. Rats received minocycline doses as 30-min intravenous infusions. In the rat pneumonia model, six clinical isolates of A. baumannii with MICs ranging from 0.03 to 4 mg/liter were studied. In this model, minocycline produced a bacteriostatic effect with a free 24-h area under the concentration-time curve (AUC)/MIC ratio of 10 to 16 and produced 1 log of bacterial killing with a free 24-h AUC/MIC of 13 to 24. These exposures can be achieved with the current FDA-approved dosage regimens of intravenous minocycline.
Keywords: Acinetobacter baumannii; minocycline; pneumonia.
Copyright © 2019 American Society for Microbiology.
Figures




Similar articles
-
Pharmacokinetics and Pharmacodynamics of Minocycline against Acinetobacter baumannii in a Neutropenic Murine Pneumonia Model.Antimicrob Agents Chemother. 2017 Apr 24;61(5):e02371-16. doi: 10.1128/AAC.02371-16. Print 2017 May. Antimicrob Agents Chemother. 2017. PMID: 28264853 Free PMC article.
-
Assessment of minocycline and polymyxin B combination against Acinetobacter baumannii.Antimicrob Agents Chemother. 2015 May;59(5):2720-5. doi: 10.1128/AAC.04110-14. Epub 2015 Feb 23. Antimicrob Agents Chemother. 2015. PMID: 25712362 Free PMC article.
-
Pharmacodynamics of minocycline against Acinetobacter baumannii studied in a pharmacokinetic model of infection.Int J Antimicrob Agents. 2017 Dec;50(6):715-717. doi: 10.1016/j.ijantimicag.2017.06.026. Epub 2017 Jul 10. Int J Antimicrob Agents. 2017. PMID: 28705678
-
Minocycline--an old drug for a new century: emphasis on methicillin-resistant Staphylococcus aureus (MRSA) and Acinetobacter baumannii.Int J Antimicrob Agents. 2009 Nov;34(5):395-401. doi: 10.1016/j.ijantimicag.2009.06.021. Epub 2009 Aug 8. Int J Antimicrob Agents. 2009. PMID: 19665876 Review.
-
Is minocycline a solution for multidrug-resistant Acinetobacter baumannii?Future Microbiol. 2014;9(3):299-305. doi: 10.2217/fmb.13.167. Future Microbiol. 2014. PMID: 24762305 Review.
Cited by
-
Intravenous Minocycline: A Review in Acinetobacter Infections.Drugs. 2016 Oct;76(15):1467-1476. doi: 10.1007/s40265-016-0636-6. Drugs. 2016. PMID: 27573640 Review.
-
Population pharmacokinetic model of oral minocycline in critically ill adult patients with ventilator-associated pneumonia.J Antimicrob Chemother. 2025 May 2;80(5):1420-1426. doi: 10.1093/jac/dkaf090. J Antimicrob Chemother. 2025. PMID: 40132622 Free PMC article.
-
Metformin reverse minocycline to inhibit minocycline-resistant Acinetobacter baumannii by destroy the outer membrane and enhance membrane potential in vitro.BMC Microbiol. 2022 Sep 12;22(1):215. doi: 10.1186/s12866-022-02629-4. BMC Microbiol. 2022. PMID: 36089583 Free PMC article.
-
Development of Modernized Acinetobacter baumannii Susceptibility Test Interpretive Criteria for Recommended Antimicrobial Agents Using Pharmacometric Approaches.Antimicrob Agents Chemother. 2023 Apr 18;67(4):e0145222. doi: 10.1128/aac.01452-22. Epub 2023 Mar 22. Antimicrob Agents Chemother. 2023. PMID: 36946729 Free PMC article.
-
Navigating Available Treatment Options for Carbapenem-Resistant Acinetobacter baumannii-calcoaceticus Complex Infections.Clin Infect Dis. 2023 May 1;76(Suppl 2):S179-S193. doi: 10.1093/cid/ciad094. Clin Infect Dis. 2023. PMID: 37125467 Free PMC article. Review.
References
-
- Camp C, Tatum OL. 2010. Review of Acinetobacter baumannii as a highly successful pathogen in times of war. Lab Med 41:6549–6657.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

