Electrospun Produced 3D Matrices for Covering of Vascular Stents: Paclitaxel Release Depending on Fiber Structure and Composition of the External Environment
- PMID: 30400260
- PMCID: PMC6265738
- DOI: 10.3390/ma11112176
Electrospun Produced 3D Matrices for Covering of Vascular Stents: Paclitaxel Release Depending on Fiber Structure and Composition of the External Environment
Abstract
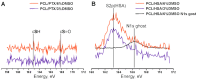
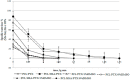
Paclitaxel is a natural, highly lipophilic anti proliferative drug widely used in medicine. We have studied the release of tritium-labeled paclitaxel (³H-PTX) from matrices destined for the coating of vascular stents and produced by the electrospinning method from the solutions of polycaprolactone (PCL) with paclitaxel (PTX) in hexafluoisopropanol (HFIP) and/or solutions of PCL with PTX and human serum albumin (HSA) in HFIP or HIFP-dimethyl sulphoxide (DMSO) blend. The release of PTX has been shown to depend on the composition of electrospinning solution, as well as the surrounding medium, particularly the concentration of free PTX and PTX-binding biomolecules present in human serum. It was shown that 3D matrices can completely release PTX without weight loss. Two-phase PTX release from optimized 3D matrices was obtained: ~27% of PTX was released in the first day, another 8% were released over the next 26 days. Wherein ~2.8%, ~2.3%, and ~0.25% of PTX was released on day 3, 9, and 27, respectively. Considering PTX toxicity, the rate of its diffusion through the arterial wall, and the data obtained the minimum cytostatic dose of the drug in the arterial wall will be maintained for at least three months.
Keywords: 3D matrix; drug release; electrospinning; paclitaxel; polycaprolactone.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures


Similar articles
-
Vascular Stents Coated with Electrospun Drug-Eluting Material: Functioning in Rabbit Iliac Artery.Polymers (Basel). 2020 Aug 4;12(8):1741. doi: 10.3390/polym12081741. Polymers (Basel). 2020. PMID: 32759856 Free PMC article.
-
Influence of Elongation of Paclitaxel-Eluting Electrospun-Produced Stent Coating on Paclitaxel Release and Transport through the Arterial Wall after Stenting.Polymers (Basel). 2021 Apr 5;13(7):1165. doi: 10.3390/polym13071165. Polymers (Basel). 2021. PMID: 33916436 Free PMC article.
-
Sirolimus-Eluting Electrospun-Produced Matrices as Coatings for Vascular Stents: Dependence of Drug Release on Matrix Structure and Composition of the External Environment.Materials (Basel). 2020 Jun 12;13(12):2692. doi: 10.3390/ma13122692. Materials (Basel). 2020. PMID: 32545664 Free PMC article.
-
An exploratory research on antitumor effect of drug-eluting slow-releasing electrospinning membranes.Heliyon. 2023 Sep 20;9(10):e20295. doi: 10.1016/j.heliyon.2023.e20295. eCollection 2023 Oct. Heliyon. 2023. PMID: 37822614 Free PMC article.
-
Aqueous N,N-diethylnicotinamide (DENA) solution as a medium for accelerated release study of paclitaxel.J Biomater Sci Polym Ed. 2004;15(4):527-42. doi: 10.1163/156856204323005343. J Biomater Sci Polym Ed. 2004. PMID: 15212332
Cited by
-
Vascular Stents Coated with Electrospun Drug-Eluting Material: Functioning in Rabbit Iliac Artery.Polymers (Basel). 2020 Aug 4;12(8):1741. doi: 10.3390/polym12081741. Polymers (Basel). 2020. PMID: 32759856 Free PMC article.
-
Porous Alginate Scaffolds Assembled Using Vaterite CaCO3 Crystals.Micromachines (Basel). 2019 May 29;10(6):357. doi: 10.3390/mi10060357. Micromachines (Basel). 2019. PMID: 31146472 Free PMC article. Review.
-
Influence of Elongation of Paclitaxel-Eluting Electrospun-Produced Stent Coating on Paclitaxel Release and Transport through the Arterial Wall after Stenting.Polymers (Basel). 2021 Apr 5;13(7):1165. doi: 10.3390/polym13071165. Polymers (Basel). 2021. PMID: 33916436 Free PMC article.
-
Bioengineering the Oxygen-Deprived Tumor Microenvironment Within a Three-Dimensional Platform for Studying Tumor-Immune Interactions.Front Bioeng Biotechnol. 2020 Sep 4;8:1040. doi: 10.3389/fbioe.2020.01040. eCollection 2020. Front Bioeng Biotechnol. 2020. PMID: 33015012 Free PMC article.
-
Sirolimus-Eluting Electrospun-Produced Matrices as Coatings for Vascular Stents: Dependence of Drug Release on Matrix Structure and Composition of the External Environment.Materials (Basel). 2020 Jun 12;13(12):2692. doi: 10.3390/ma13122692. Materials (Basel). 2020. PMID: 32545664 Free PMC article.
References
LinkOut - more resources
Full Text Sources

