Low-dose aspirin in the prevention of pre-eclampsia in China (APPEC study): protocol for a multicentre randomized controlled trial
- PMID: 30400937
- PMCID: PMC6218975
- DOI: 10.1186/s13063-018-2970-3
Low-dose aspirin in the prevention of pre-eclampsia in China (APPEC study): protocol for a multicentre randomized controlled trial
Abstract
Background: Low-dose aspirin (LDA) has been proposed as a safe and inexpensive prophylactic agent. Studies in European/Western populations have shown promising results indicating that LDA can reduce the occurrence of pre-eclampsia (PE) in women with identifiable risk factors. However, few controlled trials, particularly large randomized controlled trials, have been performed in Asian populations. The aim of this project is to evaluate the effect of LDA for PE prevention on high-risk pregnant women in China, where PE is highly prevalent and the LDA supply status is commonly suboptimal.
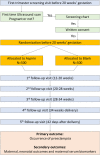
Methods/design: An open-label, multicentre randomized controlled trial is being conducted at 13 tertiary hospitals in 11 provinces in China. A total of 1000 eligible women with high-risk factors for developing PE according to their medical histories are being randomized into two groups: a control group (n = 500) and an intervention group (n = 500). Women with high-risk factors, such as a history of PE, chronic hypertension, type 1 or 2 diabetes, advanced maternal age, obesity, family history of PE or nulliparity are eligible. The control group is advised to undergo routine examinations, whereas the intervention group undergoes the routine examinations and receives LDA. LDA (100 mg/d) should be prescribed at night, initiating from early pregnancy (12-20 weeks of gestation) and lasting until 34 weeks of gestation. Demographic data and clinical endpoint outcomes, as well as biological samples (e.g., maternal blood, cord blood, amniotic fluid and placental samples), will be collected. The primary outcome is the occurrence of PE, and the secondary outcomes include maternal and neonatal outcomes and maternal biomarker levels.
Discussion: This is the first and largest multicentre randomized controlled trial to assess the effect of LDA in preventing PE in a Chinese population. The results will potentially influence the prenatal care recommendations in China regarding intervention with LDA for PE.
Trial registration: ClinicalTrials.gov, NCT02797249 . Registered on 7 June 2016.
Keywords: China; Low-dose aspirin; Pre-eclampsia; Pregnancy; Prevention.
Conflict of interest statement
Ethics approval and consent to participate
This study was reviewed and approved by the Ethics Committee of the Peking University First Hospital (reference number 2016 [1109]). Verbal and written information regarding informed consent will be presented to the caregivers and/or patients. Any modifications to the protocol that may affect the conduct of the study will be presented to the committee. The trial was registered with ClinicaTrials.gov (NCT02797249) prior to the inclusion of the first patient. All important changes to the protocol will be implemented in the trial registration. All participants will provide written informed content, and the ethics committee has approved the procedures. The full protocol will be available freely owing to the open access nature of the publication. The findings of this RCT will be submitted to a peer-reviewed journal. Abstracts will be submitted to relevant national and international conferences. The standards delineated in the guidelines of the Consolidated Standards of Reporting Trials will be followed in this RCT. All investigators will have access to the final trial dataset. We plan to communicate the trial results to the participants, healthcare professionals, policy makers, the funding agencies, the public and other relevant groups via conferences, publications and/or other data-sharing arrangements.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures

References
-
- Souza JP, Gulmezoglu AM, Vogel J, Carroli G, Lumbiganon P, Qureshi Z, et al. Moving beyond essential interventions for reduction of maternal mortality (the WHO Multicountry Survey on Maternal and Newborn Health): a cross-sectional study. Lancet. 2013;381:1747–1755. doi: 10.1016/S0140-6736(13)60686-8. - DOI - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

