Exploring Seipin: From Biochemistry to Bioinformatics Predictions
- PMID: 30402103
- PMCID: PMC6192094
- DOI: 10.1155/2018/5207608
Exploring Seipin: From Biochemistry to Bioinformatics Predictions
Abstract
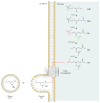
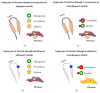
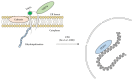
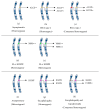
Seipin is a nonenzymatic protein encoded by the BSCL2 gene. It is involved in lipodystrophy and seipinopathy diseases. Named in 2001, all seipin functions are still far from being understood. Therefore, we reviewed much of the research, trying to find a pattern that could explain commonly observed features of seipin expression disorders. Likewise, this review shows how this protein seems to have tissue-specific functions. In an integrative view, we conclude by proposing a theoretical model to explain how seipin might be involved in the triacylglycerol synthesis pathway.
Figures






Similar articles
-
Function of seipin: new insights from Bscl2/seipin knockout mouse models.Biochimie. 2014 Jan;96:166-72. doi: 10.1016/j.biochi.2013.06.022. Epub 2013 Jul 2. Biochimie. 2014. PMID: 23831461 Review.
-
Overexpression of a short human seipin/BSCL2 isoform in mouse adipose tissue results in mild lipodystrophy.Am J Physiol Endocrinol Metab. 2012 Mar 15;302(6):E705-13. doi: 10.1152/ajpendo.00237.2011. Epub 2012 Jan 10. Am J Physiol Endocrinol Metab. 2012. PMID: 22234369
-
[Seipin/BSCL2-related motor neuron disease: Seipinopathy is a novel conformational disease associated with endoplasmic reticulum stress].Rinsho Shinkeigaku. 2007 Jun;47(6):329-35. Rinsho Shinkeigaku. 2007. PMID: 17633104 Review. Japanese.
-
Alleviation of seipinopathy-related ER stress by triglyceride storage.Hum Mol Genet. 2013 Mar 15;22(6):1157-66. doi: 10.1093/hmg/dds523. Epub 2012 Dec 18. Hum Mol Genet. 2013. PMID: 23250914
-
The expression of SEIPIN in the mouse central nervous system.Brain Struct Funct. 2016 Nov;221(8):4111-4127. doi: 10.1007/s00429-015-1151-3. Epub 2015 Nov 30. Brain Struct Funct. 2016. PMID: 26621102
Cited by
-
Discovering metabolic disease gene interactions by correlated effects on cellular morphology.Mol Metab. 2019 Jun;24:108-119. doi: 10.1016/j.molmet.2019.03.001. Epub 2019 Mar 13. Mol Metab. 2019. PMID: 30940487 Free PMC article.
-
Oligomers of the lipodystrophy protein seipin may co-ordinate GPAT3 and AGPAT2 enzymes to facilitate adipocyte differentiation.Sci Rep. 2020 Feb 24;10(1):3259. doi: 10.1038/s41598-020-59982-5. Sci Rep. 2020. PMID: 32094408 Free PMC article.
-
Seipin overexpression attenuates cerebral ischemia-reperfusion injury via preventing apoptosis and autophagy.Brain Behav. 2023 Dec;13(12):e3195. doi: 10.1002/brb3.3195. Epub 2023 Oct 27. Brain Behav. 2023. PMID: 37897134 Free PMC article.
-
Impaired mRNA splicing and proteostasis in preadipocytes in obesity-related metabolic disease.Elife. 2021 Sep 21;10:e65996. doi: 10.7554/eLife.65996. Elife. 2021. PMID: 34545810 Free PMC article.
-
SEIPIN: A Key Factor for Nuclear Lipid Droplet Generation and Lipid Homeostasis.Int J Mol Sci. 2020 Nov 2;21(21):8208. doi: 10.3390/ijms21218208. Int J Mol Sci. 2020. PMID: 33147895 Free PMC article. Review.
References
-
- Lima J. G., Nobrega L. H., de Lima N. N., do Nascimento Santos M. G., Baracho M. F., Jeronimo S. M. Clinical and laboratory data of a large series of patients with congenital generalized lipodystrophy. Diabetology & Metabolic Syndrome. 2016;8, article 23 doi: 10.1186/s13098-016-0140-x. - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources

