An ELISA-based method for detection of rabies virus nucleoprotein-specific antibodies in human antemortem samples
- PMID: 30403742
- PMCID: PMC6221316
- DOI: 10.1371/journal.pone.0207009
An ELISA-based method for detection of rabies virus nucleoprotein-specific antibodies in human antemortem samples
Abstract
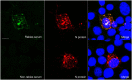
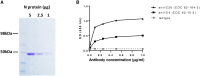
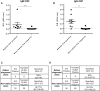
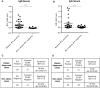
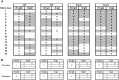
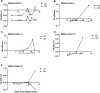
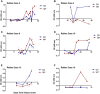
Rabies is a fatal encephalitic disease in humans and animals caused by lyssaviruses, most commonly rabies virus (RABV). Human antemortem diagnosis of rabies is a complex process involving multiple sample types and tests for the detection of antibodies, antigen (protein), and nucleic acids (genomic RNA). Serological diagnosis of human rabies includes the detection of either neutralizing or binding antibodies in the cerebrospinal fluid (CSF) or serum samples from unimmunized individuals without prior rabies vaccination or passive immunization with purified immunoglobulins. While neutralizing antibodies are targeted against the surface-expressed glycoprotein (G protein), binding antibodies to viral antigens are predominantly against the nucleoprotein (N protein), although there can be antibodies against all RABV-expressed proteins. To determine N protein-specific antibody responses in the CSF and serum during RABV infection, we developed an enzyme-linked immunosorbent assay (ELISA) with purified recombinant N protein expressed in E. coli. N protein-specific immunoglobulin (Ig) subtypes IgG and IgM were detected in the CSF or serum of previously diagnosed human rabies cases. In addition, anti-N protein seroconversion was demonstrated over the course of illness in individual rabies cases. We compared the N protein ELISA results to those of an indirect fluorescent antibody (IFA) test, the current binding antibody assay used in diagnosis, and show that our ELISA is consistent with the IFA test. Sensitivity and specificity of the N protein ELISA ranged from 78.38-100% and 75.76-96.77% with respect to the IFA results. Our data provide evidence for the use of an N protein ELISA as an additional option for the detection of RABV-specific IgG or IgM antibodies in human CSF or serum specimens.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
Similar articles
-
An evaluation of two commercially available ELISAs and one in-house reference laboratory ELISA for the determination of human anti-rabies virus antibodies.J Med Microbiol. 2009 Jun;58(Pt 6):806-810. doi: 10.1099/jmm.0.006064-0. J Med Microbiol. 2009. PMID: 19429758
-
Novel mass spectrometry based detection and identification of variants of rabies virus nucleoprotein in infected brain tissues.PLoS Negl Trop Dis. 2018 Dec 14;12(12):e0006984. doi: 10.1371/journal.pntd.0006984. eCollection 2018 Dec. PLoS Negl Trop Dis. 2018. PMID: 30550539 Free PMC article.
-
Development and evaluation of antibody-capture immunoassays for detection of Lassa virus nucleoprotein-specific immunoglobulin M and G.PLoS Negl Trop Dis. 2018 Mar 29;12(3):e0006361. doi: 10.1371/journal.pntd.0006361. eCollection 2018 Mar. PLoS Negl Trop Dis. 2018. PMID: 29596412 Free PMC article.
-
Challenges of Rabies Serology: Defining Context of Interpretation.Viruses. 2021 Jul 31;13(8):1516. doi: 10.3390/v13081516. Viruses. 2021. PMID: 34452381 Free PMC article. Review.
-
[Significance of modern methods for laboratory detection of rabies agents and identification of the zoonose immunological survey].Vopr Virusol. 2014 May-Jun;59(3):5-10. Vopr Virusol. 2014. PMID: 25335412 Review. Russian.
Cited by
-
Validation of serum apolipoprotein A1 in rabies virus-infected mice as a biomarker for the preclinical diagnosis of rabies.Microbiol Immunol. 2021 Oct;65(10):438-448. doi: 10.1111/1348-0421.12929. Epub 2021 Aug 3. Microbiol Immunol. 2021. PMID: 34270107 Free PMC article.
-
Evaluation of In-House ELISA for Antirabies Antibodies Detection in Domestic Canine.Vet Med Int. 2023 Jan 25;2023:4096258. doi: 10.1155/2023/4096258. eCollection 2023. Vet Med Int. 2023. PMID: 36743706 Free PMC article.
-
Evaluating Rabies Test Accuracy: A Systematic Review and Meta-Analysis of Human and Canine Diagnostic Methods.Diagnostics (Basel). 2025 Feb 8;15(4):412. doi: 10.3390/diagnostics15040412. Diagnostics (Basel). 2025. PMID: 40002563 Free PMC article. Review.
-
Looking at the Pathogenesis of the Rabies Lyssavirus Strain Pasteur Vaccins through a Prism of the Disorder-Based Bioinformatics.Biomolecules. 2022 Oct 7;12(10):1436. doi: 10.3390/biom12101436. Biomolecules. 2022. PMID: 36291645 Free PMC article.
-
Characterisation of a Live-Attenuated Rabies Virus Expressing a Secreted scFv for the Treatment of Rabies.Viruses. 2023 Jul 31;15(8):1674. doi: 10.3390/v15081674. Viruses. 2023. PMID: 37632016 Free PMC article.
References
-
- Lyles DS, Rupprecht CE. Rhabdoviridae In: Knipe DM, Howley PM, editors. Fields' Virology. 1 Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins; 2007. p. 1361–408.
-
- Jackson AC, Wunner WH. Rabies. Amsterdam; Boston: Elsevier/Academic Press; 2007.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

