Microfluidic Approaches for Manipulating, Imaging, and Screening C. elegans
- PMID: 30404296
- PMCID: PMC6190076
- DOI: 10.3390/mi7070123
Microfluidic Approaches for Manipulating, Imaging, and Screening C. elegans
Abstract
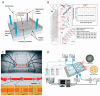
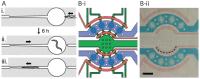
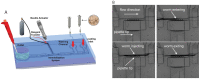
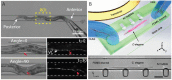
The nematode C. elegans (worm) is a small invertebrate animal widely used in studies related to fundamental biological processes, disease modelling, and drug discovery. Due to their small size and transparent body, these worms are highly suitable for experimental manipulations. In recent years several microfluidic devices and platforms have been developed to accelerate worm handling, phenotypic studies and screens. Here we review major tools and briefly discuss their usage in C. elegans research.
Keywords: C. elegans; drug discovery; electrotaxis; high-throughput screening; live imaging; microfluidics; neurobiology.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Parallel-Channel Electrotaxis and Neuron Screening of Caenorhabditis elegans.Micromachines (Basel). 2020 Aug 4;11(8):756. doi: 10.3390/mi11080756. Micromachines (Basel). 2020. PMID: 32759767 Free PMC article.
-
Detecting and Trapping of a Single C. elegans Worm in a Microfluidic Chip for Automated Microplate Dispensing.SLAS Technol. 2017 Aug;22(4):431-436. doi: 10.1177/2211068216669688. Epub 2016 Sep 26. SLAS Technol. 2017. PMID: 27630097
-
Microfluidic Technologies for High Throughput Screening Through Sorting and On-Chip Culture of C. elegans.Molecules. 2019 Nov 25;24(23):4292. doi: 10.3390/molecules24234292. Molecules. 2019. PMID: 31775328 Free PMC article. Review.
-
Microfluidic Devices in Advanced Caenorhabditis elegans Research.Molecules. 2016 Aug 2;21(8):1006. doi: 10.3390/molecules21081006. Molecules. 2016. PMID: 27490525 Free PMC article. Review.
-
An automated microfluidic system for screening Caenorhabditis elegans behaviors using electrotaxis.Biomicrofluidics. 2016 Feb 11;10(1):014117. doi: 10.1063/1.4941709. eCollection 2016 Jan. Biomicrofluidics. 2016. PMID: 26909123 Free PMC article.
Cited by
-
Experimental Evolution with Caenorhabditis Nematodes.Genetics. 2017 Jun;206(2):691-716. doi: 10.1534/genetics.115.186288. Genetics. 2017. PMID: 28592504 Free PMC article. Review.
-
Micro/Nano Devices for Chemical Analysis.Micromachines (Basel). 2016 Sep 9;7(9):164. doi: 10.3390/mi7090164. Micromachines (Basel). 2016. PMID: 30404336 Free PMC article.
-
Bioenergetic Health Assessment of a Single Caenorhabditis elegans from Postembryonic Development to Aging Stages via Monitoring Changes in the Oxygen Consumption Rate within a Microfluidic Device.Sensors (Basel). 2018 Jul 28;18(8):2453. doi: 10.3390/s18082453. Sensors (Basel). 2018. PMID: 30060586 Free PMC article.
-
Integrated Microfluidic Device for Drug Studies of Early C. Elegans Embryogenesis.Adv Sci (Weinh). 2018 Mar 8;5(5):1700751. doi: 10.1002/advs.201700751. eCollection 2018 May. Adv Sci (Weinh). 2018. PMID: 29876206 Free PMC article.
-
Quantifying male and female pheromone-based mate choice in Caenorhabditis nematodes using a novel microfluidic technique.PLoS One. 2017 Dec 13;12(12):e0189679. doi: 10.1371/journal.pone.0189679. eCollection 2017. PLoS One. 2017. PMID: 29236762 Free PMC article.
References
Publication types
LinkOut - more resources
Full Text Sources

