Axonal Injury Induces ATF3 in Specific Populations of Sacral Preganglionic Neurons in Male Rats
- PMID: 30405344
- PMCID: PMC6207596
- DOI: 10.3389/fnins.2018.00766
Axonal Injury Induces ATF3 in Specific Populations of Sacral Preganglionic Neurons in Male Rats
Abstract
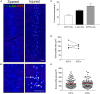
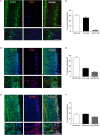
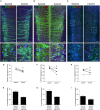
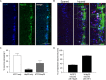
Compared to other neurons of the central nervous system, autonomic preganglionic neurons are unusual because most of their axon lies in the periphery. These axons are vulnerable to injury during surgical procedures, yet in comparison to peripheral neurons and somatic motor neurons, the impact of injury on preganglionic neurons is poorly understood. Here, we have investigated the impact of axotomy on sacral preganglionic neurons, a functionally diverse group of neurons required for micturition, defecation, and sexual function. We have previously observed that after axotomy, the injury-related transcription factor activating transcription factor-3 (ATF3) is upregulated in only half of these neurons (Peddie and Keast, 2011: PMID: 21283532). In the current study, we have investigated if this response is constrained to particular subclasses of preganglionic neurons that have specific functions or signaling properties. Seven days after unilateral pelvic nerve transection, we quantified sacral preganglionic neurons expressing ATF3, many but not all of which co-expressed c-Jun. This response was independent of soma size. Subclasses of sacral preganglionic neurons expressed combinations of somatostatin, calbindin, and neurokinin-1 receptor, each of which showed a similar response to injury. We also found that in contrast to thoracolumbar preganglionic neurons, the heat shock protein-25 (Hsp25) was not detected in naive sacral preganglionic neurons but was upregulated in many of these neurons after axotomy; the majority of these Hsp25 neurons expressed ATF3. Together, these studies reveal the molecular complexity of sacral preganglionic neurons and their responses to injury. The simultaneous upregulation of Hsp25 and ATF3 may indicate a distinct mechanism of regenerative capacity after injury.
Keywords: axon regeneration; axotomy; intermediolateral nucleus; parasympathetic; preganglionic neurons; spinal nerve injury.
Figures
References
-
- Averill S., Michael G. J., Shortland P. J., Leavesley R. C., King V. R., Bradbury E. J., et al. (2004). NGF and GDNF ameliorate the increase in ATF3 expression which occurs in dorsal root ganglion cells in response to peripheral nerve injury. Eur. J. Neurosci. 19 1437–1445. 10.1111/j.1460-9568.2004.03241.x - DOI - PubMed
-
- Barber R. P., Phelps P. E., Houser C. R., Crawford G. D., Salvaterra P. M., Vaughn J. E. (1984). The morphology and distribution of neurons containing choline acetyltransferase in the adult rat spinal cord: an immunocytochemical study. J. Comp. Neurol. 229 329–346. 10.1002/cne.902290305 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

