Dietary Zinc Supplementation Prevents Autism Related Behaviors and Striatal Synaptic Dysfunction in Shank3 Exon 13-16 Mutant Mice
- PMID: 30405356
- PMCID: PMC6204368
- DOI: 10.3389/fncel.2018.00374
Dietary Zinc Supplementation Prevents Autism Related Behaviors and Striatal Synaptic Dysfunction in Shank3 Exon 13-16 Mutant Mice
Abstract
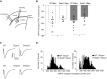
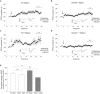
The SHANK family of synaptic proteins (SHANK1-3) are master regulators of the organizational structure of excitatory synapses in the brain. Mutations in SHANK1-3 are prevalent in patients with autism spectrum disorders (ASD), and loss of one copy of SHANK3 causes Phelan-McDermid Syndrome, a syndrome in which Autism occurs in >80% of cases. The synaptic stability of SHANK3 is highly regulated by zinc, driving the formation of postsynaptic protein complexes and increases in excitatory synaptic strength. As ASD-associated SHANK3 mutations retain responsiveness to zinc, here we investigated how increasing levels of dietary zinc could alter behavioral and synaptic deficits that occur with ASD. We performed behavioral testing together with cortico-striatal slice electrophysiology on a Shank3 -/- mouse model of ASD (Shank3 ex13-1616-/-), which displays ASD-related behaviors and structural and functional deficits at striatal synapses. We observed that 6 weeks of dietary zinc supplementation in Shank3 ex13-16-/- mice prevented ASD-related repetitive and anxiety behaviors and deficits in social novelty recognition. Dietary zinc supplementation also increased the recruitment of zinc sensitive SHANK2 to synapses, reduced synaptic transmission specifically through N-methyl-D-aspartate (NMDA)-type glutamate receptors, reversed the slowed decay tau of NMDA receptor (NMDAR)-mediated currents and occluded long term potentiation (LTP) at cortico-striatal synapses. These data suggest that alterations in NMDAR function underlie the lack of NMDAR-dependent cortico-striatal LTP and contribute to the reversal of ASD-related behaviors such as compulsive grooming. Our data reveal that dietary zinc alters neurological function from synapses to behavior, and identifies dietary zinc as a potential therapeutic agent in ASD.
Keywords: NMDA receptor; SHANK3; autism; synapse; zinc.
Figures


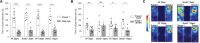
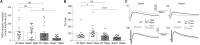
Similar articles
-
Influence of maternal zinc supplementation on the development of autism-associated behavioural and synaptic deficits in offspring Shank3-knockout mice.Mol Brain. 2020 Aug 5;13(1):110. doi: 10.1186/s13041-020-00650-0. Mol Brain. 2020. PMID: 32758248 Free PMC article.
-
Dietary zinc supplementation rescues fear-based learning and synaptic function in the Tbr1+/- mouse model of autism spectrum disorders.Mol Autism. 2022 Mar 18;13(1):13. doi: 10.1186/s13229-022-00494-6. Mol Autism. 2022. PMID: 35303947 Free PMC article.
-
Altered Striatal Synaptic Function and Abnormal Behaviour in Shank3 Exon4-9 Deletion Mouse Model of Autism.Autism Res. 2016 Mar;9(3):350-75. doi: 10.1002/aur.1529. Epub 2015 Nov 11. Autism Res. 2016. PMID: 26559786 Free PMC article.
-
The Role of Zinc and NMDA Receptors in Autism Spectrum Disorders.Pharmaceuticals (Basel). 2022 Dec 20;16(1):1. doi: 10.3390/ph16010001. Pharmaceuticals (Basel). 2022. PMID: 36678498 Free PMC article. Review.
-
Zinc deficiency and supplementation in autism spectrum disorder and Phelan-McDermid syndrome.J Neurosci Res. 2022 Apr;100(4):970-978. doi: 10.1002/jnr.25019. Epub 2022 Feb 3. J Neurosci Res. 2022. PMID: 35114017 Review.
Cited by
-
Are Essential Trace Elements Effective in Modulation of Mental Disorders? Update and Perspectives.Biol Trace Elem Res. 2022 Mar;200(3):1032-1059. doi: 10.1007/s12011-021-02733-y. Epub 2021 Apr 27. Biol Trace Elem Res. 2022. PMID: 33904124 Review.
-
Assessing sociability using the Three-Chamber Social Interaction Test and the Reciprocal Interaction Test in a genetic mouse model of ASD.Behav Brain Funct. 2024 Sep 28;20(1):24. doi: 10.1186/s12993-024-00251-0. Behav Brain Funct. 2024. PMID: 39342245 Free PMC article.
-
Zinc Water Prevents Autism-Like Behaviors in the BTBR Mice.Biol Trace Elem Res. 2023 Oct;201(10):4779-4792. doi: 10.1007/s12011-022-03548-1. Epub 2023 Jan 5. Biol Trace Elem Res. 2023. PMID: 36602746 Free PMC article.
-
Neurodevelopmental Consequences of Dietary Zinc Deficiency: A Status Report.Biol Trace Elem Res. 2023 Dec;201(12):5616-5639. doi: 10.1007/s12011-023-03630-2. Epub 2023 Mar 25. Biol Trace Elem Res. 2023. PMID: 36964812 Review.
-
Cellular zinc metabolism and zinc signaling: from biological functions to diseases and therapeutic targets.Signal Transduct Target Ther. 2024 Jan 3;9(1):6. doi: 10.1038/s41392-023-01679-y. Signal Transduct Target Ther. 2024. PMID: 38169461 Free PMC article. Review.
References
-
- Arons M. H., Thynne C. J., Grabrucker A. M., Li D., Schoen M., Cheyne J. E., et al. . (2012). Autism associated mutations in ProSAP2/Shank3 impair synaptic transmission and neurexin-neuroligin mediated transsynaptic signaling. J. Neurosci. 32, 14966–14978. 10.1523/JNEUROSCI.2215-12.2012 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

