The Role of Sodium in Diabetic Cardiomyopathy
- PMID: 30405433
- PMCID: PMC6207851
- DOI: 10.3389/fphys.2018.01473
The Role of Sodium in Diabetic Cardiomyopathy
Abstract
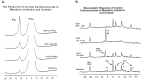
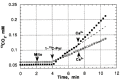
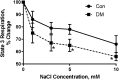
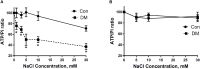
Cardiovascular complications are the major cause of mortality and morbidity in diabetic patients. The changes in myocardial structure and function associated with diabetes are collectively called diabetic cardiomyopathy. Numerous molecular mechanisms have been proposed that could contribute to the development of diabetic cardiomyopathy and have been studied in various animal models of type 1 or type 2 diabetes. The current review focuses on the role of sodium (Na+) in diabetic cardiomyopathy and provides unique data on the linkage between Na+ flux and energy metabolism, studied with non-invasive 23Na, and 31P-NMR spectroscopy, polarography, and mass spectroscopy. 23Na NMR studies allow determination of the intracellular and extracellular Na+ pools by splitting the total Na+ peak into two resonances after the addition of a shift reagent to the perfusate. Using this technology, we found that intracellular Na+ is approximately two times higher in diabetic cardiomyocytes than in control possibly due to combined changes in the activity of Na+-K+ pump, Na+/H+ exchanger 1 (NHE1) and Na+-glucose cotransporter. We hypothesized that the increase in Na+ activates the mitochondrial membrane Na+/Ca2+ exchanger, which leads to a loss of intramitochondrial Ca2+, with a subsequent alteration in mitochondrial bioenergetics and function. Using isolated mitochondria, we showed that the addition of Na+ (1-10 mM) led to a dose-dependent decrease in oxidative phosphorylation and that this effect was reversed by providing extramitochondrial Ca2+ or by inhibiting the mitochondrial Na+/Ca2+ exchanger with diltiazem. Similar experiments with 31P-NMR in isolated superfused mitochondria embedded in agarose beads showed that Na+ (3-30 mM) led to significantly decreased ATP levels and that this effect was stronger in diabetic rats. These data suggest that in diabetic cardiomyocytes, increased Na+ leads to abnormalities in oxidative phosphorylation and a subsequent decrease in ATP levels. In support of these data, using 31P-NMR, we showed that the baseline β-ATP and phosphocreatine (PCr) were lower in diabetic cardiomyocytes than in control, suggesting that diabetic cardiomyocytes have depressed bioenergetic function. Thus, both altered intracellular Na+ levels and bioenergetics and their interactions may significantly contribute to the pathology of diabetic cardiomyopathy.
Keywords: NMRS; calcium–sodium exchanger; mitochondrial bioenergetics; oxygen consumption; sodium.
Figures


Similar articles
-
Na+ effects on mitochondrial respiration and oxidative phosphorylation in diabetic hearts.Exp Biol Med (Maywood). 2001 Jun;226(6):543-51. doi: 10.1177/153537020122600606. Exp Biol Med (Maywood). 2001. PMID: 11395924
-
Metabolic control of sodium transport in streptozotocin-induced diabetic rat hearts.Biochemistry (Mosc). 2000 Apr;65(4):502-8. Biochemistry (Mosc). 2000. PMID: 10810190
-
SGLT2 inhibitor ertugliflozin decreases elevated intracellular sodium, and improves energetics and contractile function in diabetic cardiomyopathy.Biomed Pharmacother. 2023 Apr;160:114310. doi: 10.1016/j.biopha.2023.114310. Epub 2023 Jan 31. Biomed Pharmacother. 2023. PMID: 36731341 Free PMC article.
-
Cardiomyopathy associated with noninsulin-dependent diabetes.Mol Cell Biochem. 1991 Sep 18;107(1):1-20. doi: 10.1007/BF02424571. Mol Cell Biochem. 1991. PMID: 1664489 Review.
-
Intracellular calcium levels are unchanged in the diabetic heart.Cardiovasc Res. 1997 Apr;34(1):91-8. doi: 10.1016/s0008-6363(97)00034-5. Cardiovasc Res. 1997. PMID: 9217877 Review.
Cited by
-
Crosstalk between Sodium-Glucose Cotransporter Inhibitors and Sodium-Hydrogen Exchanger 1 and 3 in Cardiometabolic Diseases.Int J Mol Sci. 2021 Nov 24;22(23):12677. doi: 10.3390/ijms222312677. Int J Mol Sci. 2021. PMID: 34884494 Free PMC article. Review.
-
Underlying mechanisms and cardioprotective effects of SGLT2i and GLP-1Ra: insights from cardiovascular magnetic resonance.Cardiovasc Diabetol. 2024 Mar 11;23(1):94. doi: 10.1186/s12933-024-02181-7. Cardiovasc Diabetol. 2024. PMID: 38468245 Free PMC article. Review.
-
Diabetes-induced changes in cardiac voltage-gated ion channels.World J Diabetes. 2021 Jan 15;12(1):1-18. doi: 10.4239/wjd.v12.i1.1. World J Diabetes. 2021. PMID: 33520105 Free PMC article. Review.
-
Direct cardiac effects of SGLT2 inhibitors.Cardiovasc Diabetol. 2022 Mar 18;21(1):45. doi: 10.1186/s12933-022-01480-1. Cardiovasc Diabetol. 2022. PMID: 35303888 Free PMC article. Review.
-
Diabetic Cardiomyopathy and Ischemic Heart Disease: Prevention and Therapy by Exercise and Conditioning.Int J Mol Sci. 2020 Apr 21;21(8):2896. doi: 10.3390/ijms21082896. Int J Mol Sci. 2020. PMID: 32326182 Free PMC article. Review.
References
-
- Avkiran M. (1999). Rational basis for use of sodium-hydrogen exchange inhibitors in myocardial ischemia. Am. J. Cardiol. 83 10G–17G; discussion 17G–18G. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

