Supplementation With 2'-FL and scGOS/lcFOS Ameliorates Rotavirus-Induced Diarrhea in Suckling Rats
- PMID: 30406046
- PMCID: PMC6205980
- DOI: 10.3389/fcimb.2018.00372
Supplementation With 2'-FL and scGOS/lcFOS Ameliorates Rotavirus-Induced Diarrhea in Suckling Rats
Abstract
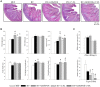
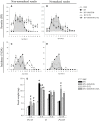
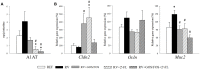
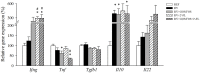
Rotavirus (RV) is considered to be the most common cause of gastroenteritis among infants aged less than 5 years old. Human milk bioactive compounds have the ability to modulate the diarrheic process caused by several intestinal pathogens. This study aimed to evaluate the potential protective role of a specific human milk oligosaccharide, 2'-fucosyllactose (2'-FL), a mixture of the prebiotic short-chain galacto-oligosaccharides and long-chain fructo-oligosaccharides 9:1 (GOS/FOS) and their combination (2'-FL+GOS/FOS) on RV-induced diarrhea in suckling rats. The nutritional intervention was performed from the second to the sixteenth day of life by oral gavage and on day 5 an RV strain was orally administered to induce infection. Fecal samples were scored daily to assess the clinical pattern of severity, incidence and duration of diarrhea. Blood and tissues were obtained at day 8 and 16 in order to evaluate the effects on the epithelial barrier and the mucosal and systemic immune responses. In the assessment of severity, incidence and duration of diarrhea, both 2'-FL and GOS/FOS displayed a beneficial effect in terms of amelioration. However, the mechanisms involved seemed to differ: 2'-FL displayed a direct ability to promote intestinal maturation and to enhance neonatal immune responses, while GOS/FOS induced an intestinal trophic effect and an RV-blocking action. The combination of 2'-FL and GOS/FOS showed additive effects in some variables. Therefore, it could be a good strategy to add these compounds in combination to infant formulas, to protect against human RV-induced diarrhea in children.
Keywords: 2′-FL; diarrhea; prebiotic; rotavirus; scGOS/lcFOS; suckling rats.
Figures

References
-
- Akbari P., Fink-Gremmels J., Willems R. H. A. M., Difilippo E., Schols H. A., Schoterman M. H. C., et al. (2017). Characterizing microbiota-independent effects of oligosaccharides on intestinal epithelial cells: insight into the role of structure and size. Structure–activity relationships of non-digestible oligosaccharides. Eur. J. Nutr. 56, 1919–1930. 10.1007/s00394-016-1234-9 - DOI - PMC - PubMed
-
- Arévalo Sureda E. A., Weström B., Pierzynowski S. G., Prykhodko O. (2017). Maturation of the intestinal epithelial barrier in neonatal rats coincides with decreased FcRn expression, replacement of vacuolated enterocytes and changed blimp-1 expression. PLoS ONE 11:e0164775. 10.1371/journal.pone.0164775 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

