Emodin Rescued Hyperhomocysteinemia-Induced Dementia and Alzheimer's Disease-Like Features in Rats
- PMID: 30407508
- PMCID: PMC6313134
- DOI: 10.1093/ijnp/pyy090
Emodin Rescued Hyperhomocysteinemia-Induced Dementia and Alzheimer's Disease-Like Features in Rats
Abstract
Background: Hyperhomocysteinemia is an independent risk factor for dementia, including Alzheimer's disease. Lowering homocysteine levels with folic acid treatment with or without vitamin B12 has shown few clinical benefits on cognition.
Methods: To verify the effect of emodin, a naturally active compound from Rheum officinale, on hyperhomocysteinemia-induced dementia, rats were treated with homocysteine injection (HCY, 400 μg/kg/d, 2 weeks) via vena caudalis. Afterwards, HCY rats with cognitive deficits were administered intragastric emodin at different concentrations for 2 weeks: 0 (HCY-E0), 20 (HCY-E20), 40 (HCY-E40), and 80 mg/kg/d (HCY-E80).
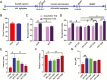
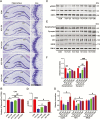
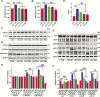
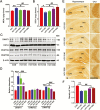
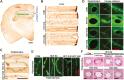
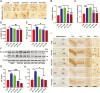
Results: β-Amyloid overproduction, tau hyperphosphorylation, and losses of neuron and synaptic proteins were detected in the hippocampi of HCY-E0 rats with cognitive deficits. HCY-E40 and HCY-E80 rats had better behavioral performance. Although it did not reduce the plasma homocysteine level, emodin (especially 80 mg/kg/d) reduced the levels of β-amyloid and tau phosphorylation, decreased the levels of β-site amyloid precursor protein-cleaving enzyme 1, and improved the activity of protein phosphatase 2A. In the hippocampi of HCY-E40 and HCY-E80 rats, the neuron numbers, levels of synaptic proteins, and phosphorylation of the cAMP responsive element-binding protein at Ser133 were increased. In addition, depressed microglial activation and reduced levels of 5-lipoxygenase, interleukin-6, and tumor necrosis factor α were also observed. Lastly, hyperhomocysteinemia-induced microangiopathic alterations, oxidative stress, and elevated DNA methyltransferases 1 and 3β were rescued by emodin.
Conclusions: Emodin represents a novel potential candidate agent for hyperhomocysteinemia-induced dementia and Alzheimer's disease-like features.
Figures
Similar articles
-
Folic acid and S-adenosylmethionine reverse Homocysteine-induced Alzheimer's disease-like pathological changes in rat hippocampus by modulating PS1 and PP2A methylation levels.Brain Res. 2024 Oct 15;1841:149095. doi: 10.1016/j.brainres.2024.149095. Epub 2024 Jun 23. Brain Res. 2024. PMID: 38917878
-
Ligustrazine Attenuates Hyperhomocysteinemia-induced Alzheimer-like Pathologies in Rats.Curr Med Sci. 2021 Jun;41(3):548-554. doi: 10.1007/s11596-021-2379-1. Epub 2021 Jun 25. Curr Med Sci. 2021. PMID: 34169425
-
Ginkgo biloba Extract EGb761 Attenuates Hyperhomocysteinemia-induced AD Like Tau Hyperphosphorylation and Cognitive Impairment in Rats.Curr Alzheimer Res. 2018;15(1):89-99. doi: 10.2174/1567205014666170829102135. Curr Alzheimer Res. 2018. PMID: 28847282
-
Homocysteine, hyperhomocysteinemia and vascular contributions to cognitive impairment and dementia (VCID).Biochim Biophys Acta. 2016 May;1862(5):1008-17. doi: 10.1016/j.bbadis.2015.11.015. Epub 2015 Dec 9. Biochim Biophys Acta. 2016. PMID: 26689889 Free PMC article. Review.
-
Hyperhomocysteinemia as a risk factor for the neuronal system disorders.J Physiol Pharmacol. 2014 Feb;65(1):15-23. J Physiol Pharmacol. 2014. PMID: 24622826 Review.
Cited by
-
A network pharmacology approach to uncover the key ingredients in Ginkgo Folium and their anti-Alzheimer's disease mechanisms.Aging (Albany NY). 2021 Jul 27;13(14):18993-19012. doi: 10.18632/aging.203348. Epub 2021 Jul 27. Aging (Albany NY). 2021. PMID: 34315132 Free PMC article.
-
Assessment of Neurodegenerative Changes in Turkeys Fed Diets with Different Proportions of Arginine and Methionine Relative to Lysine.Animals (Basel). 2022 Jun 14;12(12):1535. doi: 10.3390/ani12121535. Animals (Basel). 2022. PMID: 35739872 Free PMC article.
-
Key Phytochemicals and Biological Functions of Chuanxiong Rhizoma Against Ischemic Stroke: A Network Pharmacology and Experimental Assessment.Front Pharmacol. 2021 Dec 21;12:758049. doi: 10.3389/fphar.2021.758049. eCollection 2021. Front Pharmacol. 2021. PMID: 34992531 Free PMC article.
-
Neuroprotective, Anti-Inflammatory and Antifibrillogenic Offerings by Emodin against Alzheimer's Dementia: A Systematic Review.ACS Omega. 2024 Feb 7;9(7):7296-7309. doi: 10.1021/acsomega.3c07178. eCollection 2024 Feb 20. ACS Omega. 2024. PMID: 38405501 Free PMC article. Review.
-
Emodin alleviates cardiac fibrosis by suppressing activation of cardiac fibroblasts via upregulating metastasis associated protein 3.Acta Pharm Sin B. 2019 Jul;9(4):724-733. doi: 10.1016/j.apsb.2019.04.003. Epub 2019 Apr 22. Acta Pharm Sin B. 2019. PMID: 31384533 Free PMC article.
References
-
- Aisen PS, Schneider LS, Sano M, Diaz-Arrastia R, van Dyck CH, Weiner MF, Bottiglieri T, Jin S, Stokes KT, Thomas RG, Thal LJ, Alzheimer Disease Cooperative Study (2008)High-dose B vitamin supplementation and cognitive decline in alzheimer disease: a randomized controlled trial. Jama 300:1774–1783. - PMC - PubMed
-
- Bang OY, Chung JW, Ryoo S, Moon GJ, Kim GM, Chung CS, Lee KH(2016)Brain microangiopathy and macroangiopathy share common risk factors and biomarkers. Atherosclerosis 246:71–77. - PubMed
-
- Bessis A, Béchade C, Bernard D, Roumier A(2007)Microglial control of neuronal death and synaptic properties. Glia 55:233–238. - PubMed
-
- Block ML, Hong JS(2007)Chronic microglial activation and progressive dopaminergic neurotoxicity. Biochem Soc Trans 35:1127–1132. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

