Effects of fructose-containing sweeteners on fructose intestinal, hepatic, and oral bioavailability in dual-catheterized rats
- PMID: 30408104
- PMCID: PMC6224110
- DOI: 10.1371/journal.pone.0207024
Effects of fructose-containing sweeteners on fructose intestinal, hepatic, and oral bioavailability in dual-catheterized rats
Abstract
Objective: Fructose is commonplace in Western diets and is consumed primarily through added sugars as sucrose or high fructose corn syrup. High consumption of fructose has been linked to the development of metabolic disorders, such as cardiovascular diseases. The majority of the harmful effects of fructose can be traced to its uncontrolled and rapid metabolism, primarily within the liver. It has been speculated that the formulation of fructose-containing sweeteners can have varying impacts on its adverse effects. Unfortunately, there is limited data supporting this hypothesis. The objective of this study was to examine the impact of different fructose-containing sweeteners on the intestinal, hepatic, and oral bioavailability of fructose.
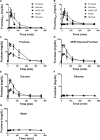
Methods: Portal and femoral vein catheters were surgically implanted in male Wistar rats. Animals were gavaged with a 1 g/kg carbohydrate solution consisting of fructose, 45% glucose/55% fructose, sucrose, glucose, or water. Blood samples were then collected from the portal and systemic circulation. Fructose levels were measured and pharmacokinetic parameters were calculated.
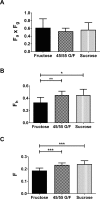
Results: Compared to animals that were gavaged with 45% glucose/55% fructose or sucrose, fructose-gavaged animals had a 40% greater fructose area under the curve and a 15% greater change in maximum fructose concentration in the portal circulation. In the systemic circulation of fructose-gavaged animals, the fructose area under the curve was 17% and 24% higher and the change in the maximum fructose concentration was 15% and 30% higher than the animals that received 45% glucose/55% fructose or sucrose, respectively. After the oral administration of fructose, 45% glucose/55% fructose, and sucrose, the bioavailability of fructose was as follows: intestinal availability was 0.62, 0.53 and 0.57; hepatic availability was 0.33, 0.45 and 0.45; and oral bioavailability was 0.19, 0.23 and 0.24, respectively.
Conclusions: Our studies show that the co-ingestion of glucose did not enhance fructose absorption, rather, it decreased fructose metabolism in the liver. The intestinal, hepatic, and oral bioavailability of fructose was similar between 45% glucose/55% fructose and sucrose.
Conflict of interest statement
CJR and CR are members of Colorado Research Partners LLC which is developing inhibitors of fructose metabolism. MTL is an inventor on the following patent applications that are related to developing inhibitors of ketohexokinase: “Plant-based inhibitors of ketohexokinase for the support of weight management” - US Patent Application #2014/0377386 A1, “Indazole Inhibitors of Fructokinase (KHK) and Methods of Use in Treating KHK-Mediated Disorders or Diseases” - US Provisional Patent Application #62/473,005, and “Botanical-Based Inhibitors, and Methods Using Same to Treat Nonalcoholic Fatty Liver Disease” - US Provisional Patent Application #62/579,372. MTL and LRV are founders of Microtek, Inc., which is a bioengineering company. These commercial entities did not provide any support for the study. This does not alter our adherence to PLOS ONE policies on sharing data and materials.
Figures


Similar articles
-
Differential Effects of Chronic Ingestion of Refined Sugars versus Natural Sweeteners on Insulin Resistance and Hepatic Steatosis in a Rat Model of Diet-Induced Obesity.Nutrients. 2020 Jul 30;12(8):2292. doi: 10.3390/nu12082292. Nutrients. 2020. PMID: 32751772 Free PMC article.
-
Eucalyptus leaf extract inhibits intestinal fructose absorption, and suppresses adiposity due to dietary sucrose in rats.Br J Nutr. 2005 Jun;93(6):957-63. doi: 10.1079/bjn20051436. Br J Nutr. 2005. PMID: 16022767
-
Impact of perinatal exposure to sucrose or high fructose corn syrup (HFCS-55) on adiposity and hepatic lipid composition in rat offspring.J Physiol. 2017 Jul 1;595(13):4379-4398. doi: 10.1113/JP274066. Epub 2017 May 26. J Physiol. 2017. PMID: 28447343 Free PMC article.
-
Fructose-containing caloric sweeteners as a cause of obesity and metabolic disorders.J Exp Biol. 2018 Mar 7;221(Pt Suppl 1):jeb164202. doi: 10.1242/jeb.164202. J Exp Biol. 2018. PMID: 29514881 Review.
-
Hepatic adaptations to sucrose and fructose.Metabolism. 2005 Sep;54(9):1189-201. doi: 10.1016/j.metabol.2005.04.004. Metabolism. 2005. PMID: 16125531 Review.
Cited by
-
The small intestine shields the liver from fructose-induced steatosis.Nat Metab. 2020 Jul;2(7):586-593. doi: 10.1038/s42255-020-0222-9. Epub 2020 Jun 22. Nat Metab. 2020. PMID: 32694791 Free PMC article.
-
13C-Stable isotope resolved metabolomics uncovers dynamic biochemical landscape of gut microbiome-host organ communications in mice.Microbiome. 2024 May 15;12(1):90. doi: 10.1186/s40168-024-01808-x. Microbiome. 2024. PMID: 38750595 Free PMC article.
-
Excess free fructose, apple juice, high fructose corn syrup and childhood asthma risk - the National Children's Study.Nutr J. 2020 Jun 23;19(1):60. doi: 10.1186/s12937-020-00578-0. Nutr J. 2020. PMID: 32576181 Free PMC article.
References
-
- Lambertz J, Weiskirchen S, Landert S, Weiskirchen R. Fructose: A Dietary Sugar in Crosstalk with Microbiota Contributing to the Development and Progression of Non-Alcoholic Liver Disease. Front Immunol. 2017;8:1159 Epub 2017/10/04. 10.3389/fimmu.2017.01159 ; PubMed Central PMCID: PMCPMC5609573. - DOI - PMC - PubMed
-
- Taskinen MR, Soderlund S, Bogl LH, Hakkarainen A, Matikainen N, Pietilainen KH, et al. Adverse effects of fructose on cardiometabolic risk factors and hepatic lipid metabolism in subjects with abdominal obesity. Journal of internal medicine. 2017;282(2):187–201. Epub 2017/05/27. 10.1111/joim.12632 . - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

