The manganese(III) porphyrin MnTnHex-2-PyP5+ modulates intracellular ROS and breast cancer cell migration: Impact on doxorubicin-treated cells
- PMID: 30408752
- PMCID: PMC6222139
- DOI: 10.1016/j.redox.2018.10.016
The manganese(III) porphyrin MnTnHex-2-PyP5+ modulates intracellular ROS and breast cancer cell migration: Impact on doxorubicin-treated cells
Abstract
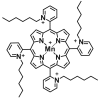
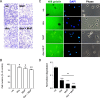
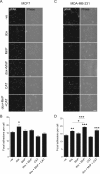
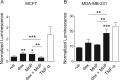
Manganese(III) porphyrins (MnPs) are superoxide dismutase (SOD) mimics with demonstrated beneficial effects in cancer treatment in combination with chemo- and radiotherapy regimens. Despite the ongoing clinical trials, little is known about the effect of MnPs on metastasis, being therefore essential to understand how MnPs affect this process. In the present work, the impact of the MnP MnTnHex-2-PyP5+ in metastasis-related processes was assessed in breast cancer cells (MCF-7 and MDA-MB-231), alone or in combination with doxorubicin (dox). The co-treatment of cells with non-cytotoxic concentrations of MnP and dox altered intracellular ROS, increasing H2O2. While MnP alone did not modify cell migration, the co-exposure led to a reduction in collective cell migration and chemotaxis. In addition, the MnP reduced the dox-induced increase in random migration of MDA-MB-231 cells. Treatment with either MnP or dox decreased the proteolytic invasion of MDA-MB-231 cells, although the effect was more pronounced upon co-exposure with both compounds. Moreover, to explore the cellular mechanisms underlying the observed effects, cell adhesion, spreading, focal adhesions, and NF-κB activation were also studied. Although differential effects were observed according to the endpoints analysed, overall, the alterations induced by MnP in dox-treated cells were consistent with a therapeutically favorable outcome.
Keywords: Cancer; Cell invasion; Cell migration; Manganese porphyrins; Redox modulation; SOD mimics.
Copyright © 2018 The Authors. Published by Elsevier B.V. All rights reserved.
Figures



References
-
- Egea J., Fabregat I., Frapart Y.M., Ghezzi P., Görlach A., Kietzmann T., Kubaichuk K., Knaus U.G., Lopez M.G., Olaso-Gonzalez G., Petry A., Schulz R., Vina J., Winyard P., Abbas K., Ademowo O.S., Afonso C.B., Andreadou I., Antelmann H., Antunes F., Aslan M., Bachschmid M.M., Barbosa R.M., Belousov V., Berndt C., Bernlohr D., Bertrán E., Bindoli A., Bottari S.P., Brito P.M., Carrara G., Casas A.I., Chatzi A., Chondrogianni N., Conrad M., Cooke M.S., Costa J.G., Cuadrado A., My-Chan Dang P., De Smet B., Debelec–Butuner B., Dias I.H.K., Dunn J.D., Edson A.J., El Assar M., El-Benna J., Ferdinandy P., Fernandes A.S., Fladmark K.E., Förstermann U., Giniatullin R., Giricz Z., Görbe A., Griffiths H., Hampl V., Hanf A., Herget J., Hernansanz-Agustín P., Hillion M., Huang J., Ilikay S., Jansen-Dürr P., Jaquet V., Joles J.A., Kalyanaraman B., Kaminskyy D., Karbaschi M., Kleanthous M., Klotz L.-O., Korac B., Korkmaz K.S., Koziel R., Kračun D., Krause K.-H., Křen V., Krieg T., Laranjinha J., Lazou A., Li H., Martínez-Ruiz A., Matsui R., McBean G.J., Meredith S.P., Messens J., Miguel V., Mikhed Y., Milisav I., Milković L., Miranda-Vizuete A., Mojović M., Monsalve M., Mouthuy P.-A., Mulvey J., Münzel T., Muzykantov V., Nguyen I.T.N., Oelze M., Oliveira N.G., Palmeira C.M., Papaevgeniou N., Pavićević A., Pedre B., Peyrot F., Phylactides M., Pircalabioru G.G., Pitt A.R., Poulsen H.E., Prieto I., Rigobello M.P., Robledinos-Antón N., Rodríguez-Mañas L., Rolo A.P., Rousset F., Ruskovska T., Saraiva N., Sasson S., Schröder K., Semen K., Seredenina T., Shakirzyanova A., Smith G.L., Soldati T., Sousa B.C., Spickett C.M., Stancic A., Stasia M.J., Steinbrenner H., Stepanić V., Steven S., Tokatlidis K., Tuncay E., Turan B., Ursini F., Vacek J., Vajnerova O., Valentová K., Van Breusegem F., Varisli L., Veal E.A., Yalçın A.S., Yelisyeyeva O., Žarković N., Zatloukalová M., Zielonka J., Touyz R.M., Papapetropoulos A., Grune T., Lamas S., Schmidt H.H.H.W., Lisa F. Di, Daiber A. European contribution to the study of ROS: a summary of the findings and prospects for the future from the cost action BM1203 (EU-ROS) Redox Biol. 2017;13:94–162. - PMC - PubMed
-
- Fernandes A.S., Saraiva N., Oliveira N.G. Redox therapeutics in breast cancer: role of SOD mimics. In: Batinic-Haberle I., Reboucas J.S., Spasojevic I., editors. Redox-Active Therapeutics. 2016. pp. 451–467.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

