Cultural adaptation and harmonization of four Nordic translations of the revised Premature Infant Pain Profile (PIPP-R)
- PMID: 30409118
- PMCID: PMC6225673
- DOI: 10.1186/s12887-018-1322-5
Cultural adaptation and harmonization of four Nordic translations of the revised Premature Infant Pain Profile (PIPP-R)
Abstract
Background: Preterm infants are especially vulnerable to pain. The intensive treatment often necessary for their survival unfortunately includes many painful interventions and procedures. Untreated pain can lead to both short- and long-term negative effects. The challenge of accurately detecting pain has been cited as a major reason for lack of pain management in these non-verbal patients. The Premature Infant Pain Profile (PIPP) is one of the most extensively validated measures for assessing procedural pain in premature infants. A revised version, PIPP-R, was recently published and is reported to be more user-friendly and precise than the original version. The aims of the study were to develop translated versions of the PIPP-R in Finnish, Icelandic, Norwegian, and Swedish languages, and to establish their content validity through a cultural adaptation process using cognitive interviews.
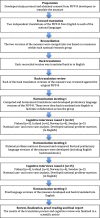
Methods: PIPP-R was translated using the recommendations from the International Society for Pharmacoeconomics and Outcomes Research and enhanced with cognitive interviews. The respondent nurse was given a copy of the translated, national version of the measure and used this together with a text describing the infant in the film to assess the pain of an infant in a short film. During the assessment the nurse was asked to verbalize her thought process (thinking aloud) and upon completion the interviewer administered probing questions (verbal probing) from a structured interview guide. The interviews were recorded, transcribed, and analyzed using a structured matrix approach.
Results: The systematic approach resulted in translated and culturally adapted versions of PIPP-R in the Finnish, Icelandic, Norwegian and Swedish languages. During the cultural adaptation process several problems were discovered regarding how the respondent understood and utilized the measure. The problems were either measure problems or other problems. Measure problems were solved by a change in the translated versions of the measure, while for other problems different solutions such as education or training were suggested.
Conclusions: This study have resulted in translations of the PIPP-R that have content validity, high degree of clinical utility and displayed beginning equivalence with each other and the original version of the measure.
Keywords: Neonatal; Pain; Pain assessment.
Conflict of interest statement
Ethics approval and consent to participate
In Sweden and Norway ethical approval was not required for this kind of study, according to the Swedish Ethical Review Act (Lag 2003:460) and the Norwegian Health Research Act of 2008. Data collection was approved by the Data Protection Officials for Research in Haukeland University Hospital (2015/14195) and at Telemark Hospital (45845) in Norway. In Finland, data collection was approved by the hospital administration at Satakunnan keskussairaala (12/2015), Pori In Iceland permission was granted by the Ethical Committee of Administrative Research at Landspitali University Hospital (01/2016) and the Icelandic Data Protection Authority was informed about details on the data collection through a formal report process (S7613/2016). In accordance with national regulations, Norwegian participants gave their written consent to participate. Consent for participation was not required in any of the other countries.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
Similar articles
-
Cultural Adaptation and Validation of the Premature Infant Pain Profile-Revised (PIPP-R) Pain Measurement Scale: Research Protocol.Int J Environ Res Public Health. 2022 Sep 28;19(19):12338. doi: 10.3390/ijerph191912338. Int J Environ Res Public Health. 2022. PMID: 36231638 Free PMC article.
-
Adaptation and Initial Validation of the Premature Infant Pain Profile-Revised (PIPP-R) in Brazil.Pain Manag Nurs. 2019 Oct;20(5):512-515. doi: 10.1016/j.pmn.2019.02.002. Epub 2019 May 15. Pain Manag Nurs. 2019. PMID: 31103519
-
The newborn infant parasympathetic evaluation index for acute procedural pain assessment in preterm infants.Pediatr Res. 2021 May;89(7):1840-1847. doi: 10.1038/s41390-020-01152-4. Epub 2020 Sep 22. Pediatr Res. 2021. PMID: 32961546
-
The premature infant pain profile: evaluation 13 years after development.Clin J Pain. 2010 Nov-Dec;26(9):813-30. doi: 10.1097/AJP.0b013e3181ed1070. Clin J Pain. 2010. PMID: 20717010 Review.
-
The premature infant and painful procedures.Pain Manag Nurs. 2000 Jun;1(2):58-65. doi: 10.1053/jpmn.2000.7781. Pain Manag Nurs. 2000. PMID: 11706460 Review.
Cited by
-
Evaluation of Pain in the Pediatric Patient Admitted to Sub-Intensive Care: What Is the Evidence? A Scoping Review.Epidemiologia (Basel). 2025 Feb 20;6(1):9. doi: 10.3390/epidemiologia6010009. Epidemiologia (Basel). 2025. PMID: 40136997 Free PMC article. Review.
-
Clinical rating scales for assessing pain in newborn infants.Cochrane Database Syst Rev. 2025 Apr 14;4(4):MR000064. doi: 10.1002/14651858.MR000064.pub2. Cochrane Database Syst Rev. 2025. PMID: 40222745
-
Adapting the Cornell assessment of pediatric delirium for Swedish context: translation, cultural validation and inter-rater reliability.BMC Pediatr. 2024 Jun 26;24(1):413. doi: 10.1186/s12887-024-04886-w. BMC Pediatr. 2024. PMID: 38926708 Free PMC article.
-
Neurophysiological and behavioral measures of pain during neonatal hip examination.Paediatr Neonatal Pain. 2019 Sep 13;1(1):15-20. doi: 10.1002/pne2.12006. eCollection 2019 Sep. Paediatr Neonatal Pain. 2019. PMID: 35546870 Free PMC article.
-
Cultural Adaptation and Validation of the Premature Infant Pain Profile-Revised (PIPP-R) Pain Measurement Scale: Research Protocol.Int J Environ Res Public Health. 2022 Sep 28;19(19):12338. doi: 10.3390/ijerph191912338. Int J Environ Res Public Health. 2022. PMID: 36231638 Free PMC article.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical