The Divergent Function of Androgen Receptor in Breast Cancer; Analysis of Steroid Mediators and Tumor Intracrinology
- PMID: 30416486
- PMCID: PMC6213369
- DOI: 10.3389/fendo.2018.00594
The Divergent Function of Androgen Receptor in Breast Cancer; Analysis of Steroid Mediators and Tumor Intracrinology
Abstract
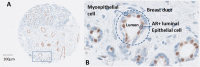
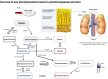
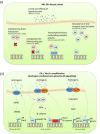
Androgen receptor (AR) is the most widely expressed steroid receptor protein in normal breast tissue and is detectable in approximately 90% of primary breast cancers and 75% of metastatic lesions. However, the role of AR in breast cancer development and progression is mired in controversy with evidence suggesting it can either inhibit or promote breast tumorigenesis. Studies have shown it to antagonize estrogen receptor alpha (ERα) DNA binding, thereby preventing pro-proliferative gene transcription; whilst others have demonstrated AR to take on the mantle of a pseudo ERα particularly in the setting of triple negative breast cancer. Evidence for a potentiating role of AR in the development of endocrine resistant breast cancer has also been mounting with reports associating high AR expression with poor response to endocrine treatment. The resurgence of interest into the function of AR in breast cancer has resulted in various emergent clinical trials evaluating anti-AR therapy and selective androgen receptor modulators in the treatment of advanced breast cancer. Trials have reported varied response rates dependent upon subtype with overall clinical benefit rates of ~19-29% for anti-androgen monotherapy, suggesting that with enhanced patient stratification AR could prove efficacious as a breast cancer therapy. Androgens and AR have been reported to facilitate tumor stemness in some cancers; a process which may be mediated through genomic or non-genomic actions of the AR, with the latter mechanism being relatively unexplored in breast cancer. Steroidogenic ligands of the AR are produced in females by the gonads and as sex-steroid precursors secreted from the adrenal glands. These androgens provide an abundant reservoir from which all estrogens are subsequently synthesized and their levels are undiminished in the event of standard hormonal therapeutic intervention in breast cancer. Steroid levels are known to be altered by lifestyle factors such as diet and exercise; understanding their potential role in dictating the function of AR in breast cancer development could therefore have wide-ranging effects in prevention and treatment of this disease. This review will outline the endogenous biochemical drivers of both genomic and non-genomic AR activation and how these may be modulated by current hormonal therapies.
Keywords: androgen receptor; breast cancer; intracrinology; non-genomic signaling; steroids.
Figures


References
-
- Picard The Role of Heat-Shock Proteins in the Regulation of Steroid Receptor Function., in Molecular Biology of Steroid and Nuclear Hormone Receptors. Progress in Gene Expression. Boston: Birkhäuser; (1998).
-
- Secreto G, Toniolo P, Berrino F, Recchione C, Cavalleri A, Pisani P, et al. . Serum and urinary androgens and risk of breast cancer in postmenopausal women. Cancer Res. (1991) 51:2572. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials

