Unraveling the journey of cancer stem cells from origin to metastasis
- PMID: 30419314
- PMCID: PMC6347501
- DOI: 10.1016/j.bbcan.2018.10.006
Unraveling the journey of cancer stem cells from origin to metastasis
Abstract
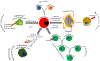
Cancer biology research over recent decades has given ample evidence for the existence of self-renewing and drug-resistant populations within heterogeneous tumors, widely recognized as cancer stem cells (CSCs). However, a lack of clear understanding about the origin, existence, maintenance, and metastatic roles of CSCs limit efforts towards the development of CSC-targeted therapy. In this review, we describe novel avenues of current CSC biology. In addition to cell fusion and horizontal gene transfer, CSCs are originated by mutations in somatic or differentiated cancer cells, resulting in de-differentiation and reprogramming. Recent studies also provided evidence for the existence of distinct or heterogeneous CSC populations within a single heterogeneous tumor. Our analysis of the literature also opens the doors for a novel hypothesis that CSC populations with specific phenotypes, metabolic profiles, and clonogenic potential metastasize to specific organs.
Keywords: Cancer stem cells; de-differentiation; metabolic reprogramming.
Copyright © 2018. Published by Elsevier B.V.
Conflict of interest statement
Conflict of interest
SKB is one of co-founders of Sanguine Diagnostics and Therapeutics, Inc. No potential conflicts of interest were disclosed by the other authors.
Figures
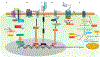
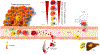
Similar articles
-
Reprogramming strategies for the establishment of novel human cancer models.Cell Cycle. 2016 Sep 16;15(18):2393-7. doi: 10.1080/15384101.2016.1196305. Epub 2016 Jun 17. Cell Cycle. 2016. PMID: 27314153 Free PMC article. Review.
-
The origin of cancer stem cells.Front Biosci (Schol Ed). 2012 Jan 1;4(3):819-30. doi: 10.2741/s302. Front Biosci (Schol Ed). 2012. PMID: 22202093 Review.
-
Cancer stem cell heterogeneity: origin and new perspectives on CSC targeting.BMB Rep. 2017 Mar;50(3):117-125. doi: 10.5483/bmbrep.2017.50.3.222. BMB Rep. 2017. PMID: 27998397 Free PMC article. Review.
-
Heterogeneity in cancer stem cells.Cancer Lett. 2015 Feb 1;357(1):63-68. doi: 10.1016/j.canlet.2014.11.040. Epub 2014 Nov 20. Cancer Lett. 2015. PMID: 25444897 Review.
-
Phenotypic subpopulations of metastatic colon cancer stem cells: genomic analysis.Cancer Genomics Proteomics. 2009 Jan-Feb;6(1):19-29. Cancer Genomics Proteomics. 2009. PMID: 19451087
Cited by
-
Using Copy Number Variation Data and Neural Networks to Predict Cancer Metastasis Origin Achieves High Area under the Curve Value with a Trade-Off in Precision.Curr Issues Mol Biol. 2024 Aug 1;46(8):8301-8319. doi: 10.3390/cimb46080490. Curr Issues Mol Biol. 2024. PMID: 39194707 Free PMC article.
-
Oncogenic LINC00698 suppresses apoptosis of melanoma stem cells to promote tumorigenesis via LINC00698-miR-3132-TCF7/hnRNPM axis.Cancer Cell Int. 2024 Jul 28;24(1):269. doi: 10.1186/s12935-024-03408-z. Cancer Cell Int. 2024. PMID: 39068483 Free PMC article.
-
Targeting Colorectal Cancer Stem Cells as an Effective Treatment for Colorectal Cancer.Technol Cancer Res Treat. 2020 Jan-Dec;19:1533033819892261. doi: 10.1177/1533033819892261. Technol Cancer Res Treat. 2020. PMID: 32748700 Free PMC article. Review.
-
Specific study of biological tumor cytology: a narrative review.Transl Cancer Res. 2021 Aug;10(8):3843-3848. doi: 10.21037/tcr-21-237. Transl Cancer Res. 2021. PMID: 35116682 Free PMC article. Review.
-
Mechanisms involved in cancer stem cell resistance in head and neck squamous cell carcinoma.Cancer Drug Resist. 2023 Feb 21;6(1):116-137. doi: 10.20517/cdr.2022.107. eCollection 2023. Cancer Drug Resist. 2023. PMID: 37065869 Free PMC article. Review.
References
-
- Makino S, The role of tumor stem-cells in regrowth of the tumor following drastic applications, Acta - Unio Internationalis Contra Cancrum 15 (Suppl. 1) (1959) 196–198. - PubMed
-
- Konrad CV, Murali R, Varghese BA, Nair R, The role of cancer stem cells in tumor heterogeneity and resistance to therapy, Can. J. Physiol. Pharmacol 95 (2017) 1–15. - PubMed
-
- Lapidot T, Sirard C, Vormoor J, A cell initiating human acute myeloid leukaemia after transplantation into SCID mice, Nature 367 (1994). - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

