α-galactosylceramide generates lung regulatory T cells through the activated natural killer T cells in mice
- PMID: 30421497
- PMCID: PMC6349240
- DOI: 10.1111/jcmm.14008
α-galactosylceramide generates lung regulatory T cells through the activated natural killer T cells in mice
Abstract
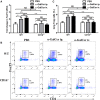
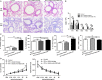
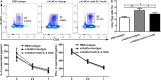
Our previous study showed that intraperitoneal injection of α-galactosylceramide (α-GalCer) has the ability to activate lung iNKT cells, but α-GalCer-activated iNKT cells do not result in airway inflammation in wild-type (WT) mice. Many studies showed that iNKT cells had the capacity to induce Treg cells, which gave rise to peripheral tolerance. Therefore, we examined the influence of intraperitoneal administration of α-GalCer on the expansion and suppressive activity of lung Treg cells using iNKT cell-knockout mice and co-culture experiments in vitro. We also compared airway inflammation and airway hyperresponsiveness (AHR) after α-GalCer administration in specific anti-CD25 mAb-treated mice. Our data showed that intraperitoneal injection of α-GalCer could promote the expansion of lung Treg cells in WT mice, but not in iNKT cell-knockout mice. However, α-GalCer administration could not boost suppressive activity of Treg cells in WT mice and iNKT cell-knockout mice. Interestingly, functional inactivation of Treg cells could induce airway inflammation and AHR in WT mice treated with α-GalCer. Furthermore, α-GalCer administration could enhance iNKT cells to secrete IL-2, and neutralization of IL-2 reduced the expansion of Treg cells in vivo and in vitro. Thus, intraperitoneal administration of α-GalCer can induce the generation of lung Treg cells in mice through the release of IL-2 by the activated iNKT cells.
Keywords: interleukin -2; invariant natural killer cells; regulatory T cells; α-galactosylceramide.
© 2018 The Authors. Journal of Cellular and Molecular Medicine published by John Wiley & Sons Ltd and Foundation for Cellular and Molecular Medicine.
Figures


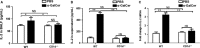
Similar articles
-
α-Galactosylceramide treatment before allergen sensitization promotes iNKT cell-mediated induction of Treg cells, preventing Th2 cell responses in murine asthma.J Biol Chem. 2019 Apr 5;294(14):5438-5455. doi: 10.1074/jbc.RA118.005418. Epub 2019 Feb 11. J Biol Chem. 2019. PMID: 30745361 Free PMC article.
-
Impact of activated invariant natural killer T cells on the expansion of regulatory T cell precursors in murine thymocytes in vitro.Immunol Lett. 2019 Feb;206:41-48. doi: 10.1016/j.imlet.2018.11.013. Epub 2018 Nov 29. Immunol Lett. 2019. PMID: 30503823
-
Regulatory T cells decrease invariant natural killer T cell-mediated pregnancy loss in mice.Mucosal Immunol. 2017 May;10(3):613-623. doi: 10.1038/mi.2016.84. Epub 2016 Oct 5. Mucosal Immunol. 2017. PMID: 27706127
-
Tailored design of NKT-stimulatory glycolipids for polarization of immune responses.J Biomed Sci. 2017 Mar 23;24(1):22. doi: 10.1186/s12929-017-0325-0. J Biomed Sci. 2017. PMID: 28335781 Free PMC article. Review.
-
Invariant natural killer T cells: innate-like T cells with potent immunomodulatory activities.Tissue Antigens. 2009 Jun;73(6):535-45. doi: 10.1111/j.1399-0039.2009.01256.x. Epub 2009 Apr 8. Tissue Antigens. 2009. PMID: 19392798 Review.
Cited by
-
Early Activation of iNKT Cells Increased Survival Time of BALB/c Mice in a Murine Model of Melioidosis.Infect Immun. 2022 Dec 15;90(12):e0026822. doi: 10.1128/iai.00268-22. Epub 2022 Nov 14. Infect Immun. 2022. PMID: 36374098 Free PMC article.
-
Natural killer T cells in allergic asthma: implications for the development of novel immunotherapeutical strategies.Front Immunol. 2024 Apr 2;15:1364774. doi: 10.3389/fimmu.2024.1364774. eCollection 2024. Front Immunol. 2024. PMID: 38629075 Free PMC article. Review.
-
Donor Treg expansion by liposomal α-galactosylceramide modulates Tfh cells and prevents sclerodermatous chronic graft-versus-host disease.Immun Inflamm Dis. 2021 Sep;9(3):721-733. doi: 10.1002/iid3.425. Epub 2021 May 4. Immun Inflamm Dis. 2021. PMID: 33942544 Free PMC article.
References
-
- Bendelac A, Savage PB, Teyton L. The biology of NKT cells. Annu Rev Immunol. 2007;25:297‐336. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

