Lysosomal Ion Channels as Decoders of Cellular Signals
- PMID: 30424907
- PMCID: PMC6340733
- DOI: 10.1016/j.tibs.2018.10.006
Lysosomal Ion Channels as Decoders of Cellular Signals
Abstract
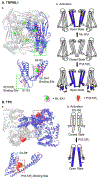
Lysosomes, the degradation center of the cell, are filled with acidic hydrolases. Lysosomes generate nutrient-sensitive signals to regulate the import of H+, hydrolases, and endocytic and autophagic cargos, as well as the export of their degradation products (catabolites). In response to environmental and cellular signals, lysosomes change their positioning, number, morphology, size, composition, and activity within minutes to hours to meet the changing cellular needs. Ion channels in the lysosome are essential transducers that mediate signal-initiated Ca2+/Fe2+/Zn2+ release and H+/Na+/K+-dependent changes of membrane potential across the perimeter membrane. Dysregulation of lysosomal ion flux impairs lysosome movement, membrane trafficking, nutrient sensing, membrane repair, organelle membrane contact, and lysosome biogenesis and adaptation. Hence, activation and inhibition of lysosomal channels by synthetic modulators may tune lysosome function to maintain cellular health and promote cellular clearance in lysosome storage disorders.
Keywords: TFEB; TPC2; TRPML1; lysosomal exocytosis; lysosomal storage disease; mTOR.
Copyright © 2018 Elsevier Ltd. All rights reserved.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

