The future of quantum biology
- PMID: 30429265
- PMCID: PMC6283985
- DOI: 10.1098/rsif.2018.0640
The future of quantum biology
Abstract
Biological systems are dynamical, constantly exchanging energy and matter with the environment in order to maintain the non-equilibrium state synonymous with living. Developments in observational techniques have allowed us to study biological dynamics on increasingly small scales. Such studies have revealed evidence of quantum mechanical effects, which cannot be accounted for by classical physics, in a range of biological processes. Quantum biology is the study of such processes, and here we provide an outline of the current state of the field, as well as insights into future directions.
Keywords: artificial photosynthesis; charge transfer; enzyme catalysis; light harvesting; quantum technology; sensing.
© 2018 The Author(s).
Conflict of interest statement
We declare we have no competing interests.
Figures
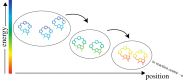
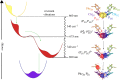
 —a high exciton state,
—a high exciton state,  —an exciton state with some charge-transfer character,
—an exciton state with some charge-transfer character,  —a charge-transfer state mixed with an exciton state, and
—a charge-transfer state mixed with an exciton state, and  —the final charge-separated state. Note that an intermediate between the last two states, involving ChlD1, is omitted. The first three steps involve coherent relaxation due to coupling with a vibrational mode, while the last step involves incoherent transfer. Symbols: D1 and D2, two branches of the symmetric reaction centre structure; Chl, chlorophyll; P, primary electron donor/special-pair Chl; Phe, pheophytin. Adapted from Romero et al. [23].
—the final charge-separated state. Note that an intermediate between the last two states, involving ChlD1, is omitted. The first three steps involve coherent relaxation due to coupling with a vibrational mode, while the last step involves incoherent transfer. Symbols: D1 and D2, two branches of the symmetric reaction centre structure; Chl, chlorophyll; P, primary electron donor/special-pair Chl; Phe, pheophytin. Adapted from Romero et al. [23].
References
-
- F'orster T. 1946. Energiewanderung und Fluoreszenz. Naturwissenschaften, 6, 166–175. ( 10.1007/BF00585226) - DOI
-
- Marcus RA. 1956. On the theory of oxidation-reduction reactions involving electron transfer. I. J. Chem. Phys. 24, 966–978. ( 10.1063/1.1742723) - DOI
-
- Bohr N. 1933. Light and life. Nature 131, 421–423. ( 10.1038/131421a0) - DOI
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
