CSF1R-related leukoencephalopathy: A major player in primary microgliopathies
- PMID: 30429277
- PMCID: PMC6329328
- DOI: 10.1212/WNL.0000000000006642
CSF1R-related leukoencephalopathy: A major player in primary microgliopathies
Abstract
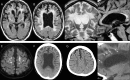
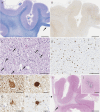
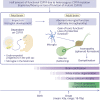
Since the discovery of CSF1R gene mutations in families with hereditary diffuse leukoencephalopathy with spheroids in 2012, more than 70 different mutations have been identified around the world. Through the analyses of mutation carriers, CSF1R-related leukoencephalopathy has been distinctly characterized clinically, radiologically, and pathologically. Typically, patients present with frontotemporal dementia-like phenotype in their 40s-50s, accompanied by motor symptoms, including pyramidal and extrapyramidal signs. Women tend to develop the clinical symptoms at a younger age than men. On brain imaging, in addition to white matter abnormalities, thinning of the corpus callosum, diffusion-restricted lesions in the white matter, and brain calcifications are hallmarks. Primary axonopathy followed by demyelination was suggested by pathology. Haploinsufficiency of colony-stimulating factor-1 receptor (CSF1R) is evident in a patient with a frameshift mutation, facilitating the establishment of Csf1r haploinsufficient mouse model. These mice develop clinical, radiologic, and pathologic phenotypes consistent with those of human patients with CSF1R mutations. In vitro, perturbation of CSF1R signaling is shown in cultured cells expressing mutant CSF1R. However, the underlying mechanisms by which CSF1R mutations selectively lead to white matter degeneration remains to be elucidated. Given that CSF1R mainly expresses in microglia, CSF1R-related leukoencephalopathy is representative of primary microgliopathies, of which microglia have a pivotal and primary role in pathogenesis. In this review, we address the current knowledge of CSF1R-related leukoencephalopathy and discuss the putative pathophysiology, with a focus on microglia, as well as future research directions.
© 2018 American Academy of Neurology.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
