Flexible elastomer patch with vertical silicon nanoneedles for intracellular and intratissue nanoinjection of biomolecules
- PMID: 30430139
- PMCID: PMC6226283
- DOI: 10.1126/sciadv.aau6972
Flexible elastomer patch with vertical silicon nanoneedles for intracellular and intratissue nanoinjection of biomolecules
Abstract
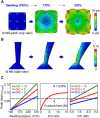
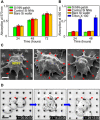
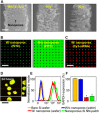
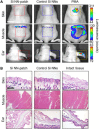
Vertically ordered arrays of silicon nanoneedles (Si NNs), due to their nanoscale dimension and low cytotoxicity, could enable minimally invasive nanoinjection of biomolecules into living biological systems such as cells and tissues. Although production of these Si NNs on a bulk Si wafer has been achieved through standard nanofabrication technology, there exists a large mismatch at the interface between the rigid, flat, and opaque Si wafer and soft, curvilinear, and optically transparent biological systems. Here, we report a unique methodology that is capable of constructing vertically ordered Si NNs on a thin layer of elastomer patch to flexibly and transparently interface with biological systems. The resulting outcome provides important capabilities to form a mechanically elastic interface between Si NNs and biological systems, and simultaneously enables direct imaging of their real-time interactions under the transparent condition. We demonstrate its utility in intracellular, intradermal, and intramuscular nanoinjection of biomolecules into various kinds of biological cells and tissues at their length scales.
Figures


Similar articles
-
Integrating Porous Silicon Nanoneedles within Medical Devices for Nucleic Acid Nanoinjection.ACS Nano. 2024 Jun 11;18(23):14938-14953. doi: 10.1021/acsnano.4c00206. Epub 2024 May 10. ACS Nano. 2024. PMID: 38726598 Free PMC article.
-
Optically transparent vertical silicon nanowire arrays for live-cell imaging.J Nanobiotechnology. 2021 Feb 17;19(1):51. doi: 10.1186/s12951-021-00795-7. J Nanobiotechnology. 2021. PMID: 33596905 Free PMC article.
-
Electroassisted transfer of vertical silicon wire arrays using a sacrificial porous silicon layer.Nano Lett. 2013 Sep 11;13(9):4362-8. doi: 10.1021/nl4021705. Epub 2013 Aug 13. Nano Lett. 2013. PMID: 23919596
-
Versatile Particle-Based Route to Engineer Vertically Aligned Silicon Nanowire Arrays and Nanoscale Pores.ACS Appl Mater Interfaces. 2015 Oct 28;7(42):23717-24. doi: 10.1021/acsami.5b07777. Epub 2015 Oct 13. ACS Appl Mater Interfaces. 2015. PMID: 26428032
-
Tutorial: using nanoneedles for intracellular delivery.Nat Protoc. 2021 Oct;16(10):4539-4563. doi: 10.1038/s41596-021-00600-7. Epub 2021 Aug 23. Nat Protoc. 2021. PMID: 34426708 Review.
Cited by
-
Interrogation on the Cellular Nano-Interface and Biosafety of Repeated Nano-Electroporation by Nanostraw System.Biosensors (Basel). 2022 Jul 13;12(7):522. doi: 10.3390/bios12070522. Biosensors (Basel). 2022. PMID: 35884325 Free PMC article.
-
Integrating Porous Silicon Nanoneedles within Medical Devices for Nucleic Acid Nanoinjection.ACS Nano. 2024 Jun 11;18(23):14938-14953. doi: 10.1021/acsnano.4c00206. Epub 2024 May 10. ACS Nano. 2024. PMID: 38726598 Free PMC article.
-
Challenges of Gene Editing Therapies for Genodermatoses.Int J Mol Sci. 2023 Jan 24;24(3):2298. doi: 10.3390/ijms24032298. Int J Mol Sci. 2023. PMID: 36768619 Free PMC article. Review.
-
Microfluidic mechanoporation for cellular delivery and analysis.Mater Today Bio. 2021 Dec 20;13:100193. doi: 10.1016/j.mtbio.2021.100193. eCollection 2022 Jan. Mater Today Bio. 2021. PMID: 35005598 Free PMC article. Review.
-
High-Aspect-Ratio Nanostructured Surfaces as Biological Metamaterials.Adv Mater. 2020 Mar;32(9):e1903862. doi: 10.1002/adma.201903862. Epub 2020 Jan 16. Adv Mater. 2020. PMID: 31944430 Free PMC article. Review.
References
-
- Shalek A. K., Robinson J. T., Karp E. S., Lee J. S., Ahn D.-R., Yoon M.-H., Sutton A., Jorgolli M., Gertner R. S., Gujral T. S., MacBeath G., Yang E. G., Park H., Vertical silicon nanowires as a universal platform for delivering biomolecules into living cells. Proc. Natl. Acad. Sci. U.S.A. 107, 1870–1875 (2010). - PMC - PubMed
-
- Yan R. X., Park J.-H., Choi Y., Heo C.-J., Yang S.-M., Lee L. P., Yang P. D., Nanowire-based single-cell endoscopy. Nat. Nanotechnol. 7, 191–196 (2012). - PubMed
-
- Luo Z., Jiang Y., Myers B. D., Isheim D., Wu J., Zimmerman J. F., Wang Z., Li Q., Wang Y., Chen X., Dravid V. P., Seidman D. N., Tian B., Atomic gold–enabled three-dimensional lithography for silicon mesostructures. Science 348, 1451–1455 (2015). - PubMed
-
- Spira M. E., Hai A., Multi-electrode array technologies for neuroscience and cardiology. Nat. Nanotechnol. 8, 83–94 (2013). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

