Enhancing Protein Stability with Genetically Encoded Noncanonical Amino Acids
- PMID: 30433771
- PMCID: PMC6426444
- DOI: 10.1021/jacs.8b07157
Enhancing Protein Stability with Genetically Encoded Noncanonical Amino Acids
Abstract
The ability to add noncanonical amino acids to the genetic code may allow one to evolve proteins with new or enhanced properties using a larger set of building blocks. To this end, we have been able to select mutant proteins with enhanced thermal properties from a library of E. coli homoserine O-succinyltransferase ( metA) mutants containing randomly incorporated noncanonical amino acids. Here, we show that substitution of Phe 21 with ( p-benzoylphenyl)alanine (pBzF), increases the melting temperature of E. coli metA by 21 °C. This dramatic increase in thermal stability, arising from a single mutation, likely results from a covalent adduct between Cys 90 and the keto group of pBzF that stabilizes the dimeric form of the enzyme. These experiments show that an expanded genetic code can provide unique solutions to the evolution of proteins with enhanced properties.
Figures

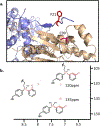
References
-
- Liu CC; Schultz PG, Adding new chemistries to the genetic code. Annu Rev Biochem 2010, 79, 413–44. - PubMed
-
- Wang L; Schultz PG, Expanding the genetic code. Angew Chem Int Ed Engl 2004, 44 (1), 34–66. - PubMed
-
- Wang L; Xie J; Schultz PG, Expanding the genetic code. Annu Rev Biophys Biomol Struct 2006, 35, 225–49. - PubMed
-
- Xie J; Schultz PG, A chemical toolkit for proteins — an expanded genetic code. Nature Reviews 2006, 7, 775–782. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

