Metformin causes cancer cell death through downregulation of p53-dependent differentiated embryo chondrocyte 1
- PMID: 30442142
- PMCID: PMC6238313
- DOI: 10.1186/s12929-018-0478-5
Metformin causes cancer cell death through downregulation of p53-dependent differentiated embryo chondrocyte 1
Abstract
Background: Metformin is the most commonly used first-line medicine for type II diabetes mellitus. Acting via AMP-activated protein kinase, it has been used for more than 60 years and has an outstanding safety record. Metformin also offers protection against cancer, but its precise mechanisms remain unclear.
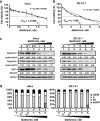
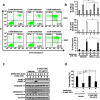
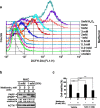
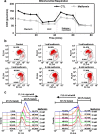
Methods: We first examined the cytotoxic effects of metformin in the HeLa human cervical carcinoma and ZR-75-1 breast cancer cell lines using assays of cell viability, cleaved poly-ADP-ribose polymerase, and Annexin V-fluorescein isothiocyanate apoptosis, as well as flow cytometric analyses of the cell cycle profile and reactive oxygen species (ROS). We later clarified the effect of metformin on p53 protein stability using transient transfection and cycloheximide chase analyses.
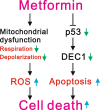
Results: We observed that metformin represses cell cycle progression, thereby inducing subG1 populations, and had induced apoptosis through downregulation of p53 protein and a target gene, differentiated embryo chondrocyte 1 (DEC1). In addition, metformin increased intracellular ROS levels, but N-acetyl cysteine, a ROS scavenger, failed to suppress metformin-induced apoptosis. Further results showed that metformin disrupted the electron transport chain and collapsed the mitochondrial membrane potential, which may be the cause of the elevated ROS levels. Examination of the mechanisms underlying metformin-induced HeLa cell death revealed that reduced stability of p53 in metformin-treated cells leads to decreases in DEC1 and induction of apoptosis.
Conclusion: The involvement of DEC1 provides new insight into the positive or negative functional roles of p53 in the metformin-induced cytotoxicity in tumor cells.
Keywords: Apoptosis; DEC1; Metformin; Reactive oxygen species; p53.
Conflict of interest statement
Ethics approval and consent to participate
“Not applicable” (The present paper did not involve the use of any animal or human data).
Consent for publication
“Not applicable”.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures


Similar articles
-
Metformin induces microRNA-34a to downregulate the Sirt1/Pgc-1α/Nrf2 pathway, leading to increased susceptibility of wild-type p53 cancer cells to oxidative stress and therapeutic agents.Free Radic Biol Med. 2014 Sep;74:21-34. doi: 10.1016/j.freeradbiomed.2014.06.010. Epub 2014 Jun 24. Free Radic Biol Med. 2014. PMID: 24970682
-
The Mechanisms Underlying the Cytotoxic Effects of Copper Via Differentiated Embryonic Chondrocyte Gene 1.Int J Mol Sci. 2019 Oct 22;20(20):5225. doi: 10.3390/ijms20205225. Int J Mol Sci. 2019. PMID: 31652494 Free PMC article.
-
Metformin Modulates Cyclin D1 and P53 Expression to Inhibit Cell Proliferation and to Induce Apoptosis in Cervical Cancer Cell Lines.Asian Pac J Cancer Prev. 2019 Jun 1;20(6):1667-1673. doi: 10.31557/APJCP.2019.20.6.1667. Asian Pac J Cancer Prev. 2019. PMID: 31244286 Free PMC article.
-
Differentiated Embryo-Chondrocyte Expressed Gene1 and Parkinson's Disease: New Insights and Therapeutic Perspectives.Curr Neuropharmacol. 2023;21(11):2251-2265. doi: 10.2174/1570159X21666230502123729. Curr Neuropharmacol. 2023. PMID: 37132111 Free PMC article. Review.
-
Role of differentiated embryo-chondrocyte expressed gene 1 (DEC1) in immunity.Int Immunopharmacol. 2022 Jan;102:107892. doi: 10.1016/j.intimp.2021.107892. Epub 2021 Jun 30. Int Immunopharmacol. 2022. PMID: 34215553 Review.
Cited by
-
Incorporation of Sulfonamide Moiety into Biguanide Scaffold Results in Apoptosis Induction and Cell Cycle Arrest in MCF-7 Breast Cancer Cells.Int J Mol Sci. 2021 May 26;22(11):5642. doi: 10.3390/ijms22115642. Int J Mol Sci. 2021. PMID: 34073245 Free PMC article.
-
The antitumor properties of metformin and phenformin reflect their ability to inhibit the actions of differentiated embryo chondrocyte 1.Cancer Manag Res. 2019 Jul 15;11:6567-6579. doi: 10.2147/CMAR.S210637. eCollection 2019. Cancer Manag Res. 2019. PMID: 31410055 Free PMC article.
-
Metformin as an adjuvant in breast cancer treatment.SAGE Open Med. 2019 Jul 16;7:2050312119865114. doi: 10.1177/2050312119865114. eCollection 2019. SAGE Open Med. 2019. PMID: 31360518 Free PMC article. Review.
-
Common Pathogenetic Mechanisms Underlying Aging and Tumor and Means of Interventions.Aging Dis. 2022 Jul 11;13(4):1063-1091. doi: 10.14336/AD.2021.1208. eCollection 2022 Jul 11. Aging Dis. 2022. PMID: 35855334 Free PMC article.
-
Molecular and Cellular Mechanisms of Metformin in Cervical Cancer.Cancers (Basel). 2021 May 22;13(11):2545. doi: 10.3390/cancers13112545. Cancers (Basel). 2021. PMID: 34067321 Free PMC article. Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

