SEPROGADIC - serum protein-based gastric cancer prediction model for prognosis and selection of proper adjuvant therapy
- PMID: 30442939
- PMCID: PMC6237900
- DOI: 10.1038/s41598-018-34858-x
SEPROGADIC - serum protein-based gastric cancer prediction model for prognosis and selection of proper adjuvant therapy
Abstract
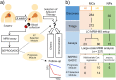
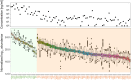
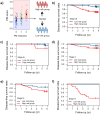
Gastric cancer (GC) patients usually receive surgical treatment. Postoperative therapeutic options such as anticancer adjuvant therapies (AT) based on prognostic prediction models would provide patient-specific treatment to decrease postsurgical morbidity and mortality rates. Relevant prognostic factors in resected GC patient's serum may improve therapeutic measures in a non-invasive manner. In order to develop a GC prognostic model, we designed a retrospective study. In this study, serum samples were collected from 227 patients at a 4-week recovery period after D2 lymph node dissection, and 103 cancer-related serum proteins were analyzed by multiple reaction monitoring mass spectrometry. Using the quantitative values of the serum proteins, we developed SEPROGADIC (SErum PROtein-based GAstric cancer preDICtor) prognostic model consisting of 6 to 14 serum proteins depending on detailed purposes of the model, prognosis prediction and proper AT selection. SEPROGADIC could clearly classify patients with good or bad prognosis at each TNM stage (1b, 2, 3 and 4) and identify a patient subgroup who would benefit from CCRT (combined chemoradiation therapy) rather than CTX (chemotherapy), or vice versa. Our study demonstrated that serum proteins could serve as prognostic factors along with clinical stage information in patients with resected gastric cancer, thus allowing patient-tailored postsurgical treatment.
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
Prognostic analysis of stage III gastric cancer after curative surgery according to the newest TNM classification.Clin Transl Oncol. 2019 Feb;21(2):232-238. doi: 10.1007/s12094-018-1913-5. Epub 2018 Jul 2. Clin Transl Oncol. 2019. PMID: 29968135
-
[Prognostic factors of lymph node-negative metastasis gastric cancer].Zhonghua Wei Chang Wai Ke Za Zhi. 2017 Feb 25;20(2):190-194. Zhonghua Wei Chang Wai Ke Za Zhi. 2017. PMID: 28226354 Chinese.
-
Clinical Outcomes and the Role of Adjuvant Concurrent Chemoradiation Therapy in D2-resected LN-positive Young Patients (≤45 Years) With Gastric Cancer.Anticancer Res. 2019 Oct;39(10):5811-5820. doi: 10.21873/anticanres.13785. Anticancer Res. 2019. PMID: 31570486
-
Pretherapeutic evaluation of patients with upper gastrointestinal tract cancer using endoscopic and laparoscopic ultrasonography.Dan Med J. 2012 Dec;59(12):B4568. Dan Med J. 2012. PMID: 23290296 Review.
-
Overview of resistance to systemic therapy in patients with breast cancer.Adv Exp Med Biol. 2007;608:1-22. doi: 10.1007/978-0-387-74039-3_1. Adv Exp Med Biol. 2007. PMID: 17993229 Review.
Cited by
-
Differential Urinary Proteome Analysis for Predicting Prognosis in Type 2 Diabetes Patients with and without Renal Dysfunction.Int J Mol Sci. 2020 Jun 14;21(12):4236. doi: 10.3390/ijms21124236. Int J Mol Sci. 2020. PMID: 32545899 Free PMC article.
-
DKK3 and SERPINB5 as novel serum biomarkers for gastric cancer: facilitating the development of risk prediction models for gastric cancer.Front Oncol. 2025 Mar 31;15:1536491. doi: 10.3389/fonc.2025.1536491. eCollection 2025. Front Oncol. 2025. PMID: 40231256 Free PMC article.
-
A predictive signature for oxaliplatin and 5-fluorouracil based chemotherapy in locally advanced gastric cancer.Transl Oncol. 2021 Jan;14(1):100901. doi: 10.1016/j.tranon.2020.100901. Epub 2020 Oct 20. Transl Oncol. 2021. PMID: 33091827 Free PMC article.
-
Plasma Protein Biomarkers Associated with Higher Ovarian Cancer Risk in BRCA1/2 Carriers.Cancers (Basel). 2021 May 11;13(10):2300. doi: 10.3390/cancers13102300. Cancers (Basel). 2021. PMID: 34064977 Free PMC article.
-
An Exploratory Pilot Study with Plasma Protein Signatures Associated with Response of Patients with Depression to Antidepressant Treatment for 10 Weeks.Biomedicines. 2020 Oct 28;8(11):455. doi: 10.3390/biomedicines8110455. Biomedicines. 2020. PMID: 33126421 Free PMC article.
References
-
- Global Burden of Disease Cancer Collaboration et al. Global, Regional, and National Cancer Incidence, Mortality, Years of Life Lost, Years Lived With Disability, and Disability-Adjusted Life-years for 32 Cancer Groups, 1990 to 2015: A Systematic Analysis for the Global Burden of Disease Study. JAMA Oncol 3, 524–548, 10.1001/jamaoncol.2016.5688 (2017). - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

