Functionalizing cell-mimetic giant vesicles with encapsulated bacterial biosensors
- PMID: 30443325
- PMCID: PMC6227772
- DOI: 10.1098/rsfs.2018.0024
Functionalizing cell-mimetic giant vesicles with encapsulated bacterial biosensors
Abstract
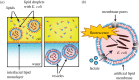
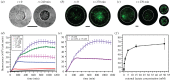
The design of vesicle microsystems as artificial cells (bottom-up synthetic biology) has traditionally relied on the incorporation of molecular components to impart functionality. These cell mimics have reduced capabilities compared with their engineered biological counterparts (top-down synthetic biology), as they lack the powerful metabolic and regulatory pathways associated with living systems. There is increasing scope for using whole intact cellular components as functional modules within artificial cells, as a route to increase the capabilities of artificial cells. In this feasibility study, we design and embed genetically engineered microbes (Escherichia coli) in a vesicle-based cell mimic and use them as biosensing modules for real-time monitoring of lactate in the external environment. Using this conceptual framework, the functionality of other microbial devices can be conferred into vesicle microsystems in the future, bridging the gap between bottom-up and top-down synthetic biology.
Keywords: artificial cells; biosensing; cellular bionics; giant lipid vesicles; microfluidics; synthetic biology.
Conflict of interest statement
The authors have no competing interests.
Figures

Similar articles
-
Constructing vesicle-based artificial cells with embedded living cells as organelle-like modules.Sci Rep. 2018 Mar 14;8(1):4564. doi: 10.1038/s41598-018-22263-3. Sci Rep. 2018. PMID: 29540757 Free PMC article.
-
Bottom-Up Assembly of Functional Intracellular Synthetic Organelles by Droplet-Based Microfluidics.Small. 2020 Jul;16(27):e1906424. doi: 10.1002/smll.201906424. Epub 2020 Feb 20. Small. 2020. PMID: 32078238
-
Measuring Encapsulation Efficiency in Cell-Mimicking Giant Unilamellar Vesicles.ACS Synth Biol. 2023 Apr 21;12(4):1227-1238. doi: 10.1021/acssynbio.2c00684. Epub 2023 Mar 28. ACS Synth Biol. 2023. PMID: 36977193 Free PMC article.
-
Microfluidics for Artificial Life: Techniques for Bottom-Up Synthetic Biology.Micromachines (Basel). 2019 Apr 30;10(5):299. doi: 10.3390/mi10050299. Micromachines (Basel). 2019. PMID: 31052344 Free PMC article. Review.
-
Microfluidic Handling and Analysis of Giant Vesicles for Use as Artificial Cells: A Review.Adv Biosyst. 2019 Jun;3(6):e1800318. doi: 10.1002/adbi.201800318. Epub 2019 May 7. Adv Biosyst. 2019. PMID: 32648705 Review.
Cited by
-
Chromatophores efficiently promote light-driven ATP synthesis and DNA transcription inside hybrid multicompartment artificial cells.Proc Natl Acad Sci U S A. 2021 Feb 16;118(7):e2012170118. doi: 10.1073/pnas.2012170118. Proc Natl Acad Sci U S A. 2021. PMID: 33526592 Free PMC article.
-
Polymer Encapsulation of Bacterial Biosensors Enables Coculture with Mammalian Cells.ACS Synth Biol. 2022 Mar 18;11(3):1303-1312. doi: 10.1021/acssynbio.1c00577. Epub 2022 Mar 4. ACS Synth Biol. 2022. PMID: 35245022 Free PMC article.
-
Membrane anchoring facilitates colocalization of enzymes in plant cytochrome P450 redox systems.Commun Biol. 2021 Sep 9;4(1):1057. doi: 10.1038/s42003-021-02604-1. Commun Biol. 2021. PMID: 34504298 Free PMC article.
-
Force Generation by Enhanced Diffusion in Enzyme-Loaded Vesicles.Nano Lett. 2025 Apr 9;25(14):5754-5761. doi: 10.1021/acs.nanolett.5c00306. Epub 2025 Mar 26. Nano Lett. 2025. PMID: 40138661 Free PMC article.
-
Chemical communication at the synthetic cell/living cell interface.Commun Chem. 2021 Nov 25;4(1):161. doi: 10.1038/s42004-021-00597-w. Commun Chem. 2021. PMID: 36697795 Free PMC article. Review.
References
Associated data
LinkOut - more resources
Full Text Sources
Miscellaneous

