Injectable Biodegradable Chitosan-Alginate 3D Porous Gel Scaffold for mRNA Vaccine Delivery
- PMID: 30444317
- PMCID: PMC6611697
- DOI: 10.1002/mabi.201800242
Injectable Biodegradable Chitosan-Alginate 3D Porous Gel Scaffold for mRNA Vaccine Delivery
Abstract
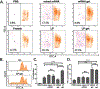
mRNA vaccines have proven to be more stable, effective, and specific than protein/peptide-based vaccines in stimulating both humoral and cellular immune response. However, mRNA's fast degradation rate and low-transfection efficiency in vivo impede its potential in vaccination. Recent research in gene delivery has focused on nonviral vaccine carriers and either implantable or injectable delivery systems to improve transgene expression in vivo. Here, an injectable chitosan-alginate gel scaffold for the local delivery of mRNA vaccines is reported. Gel scaffold biodegradation rates and biocompatibility are quantified. Scaffold-mediated mRNA in vivo transgene expression as well as ovalbumin antigen specific cellular and humoral immune responses are evaluated in vivo. Luciferase reporter protein expression resulting from mRNA lipoplex-loaded gel scaffolds is five times higher than systemic injection. Compared to systemic injections of naked mRNA or mRNA:lipoplexes, elevated levels of T cell proliferation and IFN-γ secretion are seen with in vivo scaffold-mediated mRNA lipoplex delivery. Furthermore, a humoral response (ovalbumin antigen specific IgG levels) is observed as early as week 1 for scaffold-mediated mRNA lipoplex delivery, while protein-based immunization did not elicit IgG production until 2 weeks post-injection. Results suggest that injectable scaffold mRNA vaccine delivery maybe a viable alternative to traditional nucleic acid immunization methods.
Keywords: biodegradable; injectable immunizing scaffold; mRNA vaccine delivery.
© 2018 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Conflict of interest statement
Conflict of Interest
The authors declare no conflict of interest.
Figures

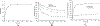

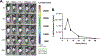
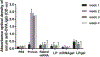
Similar articles
-
pH Sensitive phosphorylated chitosan hydrogel as vaccine delivery system for intramuscular immunization.J Biomater Appl. 2017 May;31(10):1358-1369. doi: 10.1177/0885328217704139. Epub 2017 Apr 7. J Biomater Appl. 2017. PMID: 28387574
-
Development of a lipoplex-type mRNA carrier composed of an ionizable lipid with a vitamin E scaffold and the KALA peptide for use as an ex vivo dendritic cell-based cancer vaccine.J Control Release. 2019 Sep 28;310:36-46. doi: 10.1016/j.jconrel.2019.08.002. Epub 2019 Aug 3. J Control Release. 2019. PMID: 31386869
-
Evaluation of N-phosphonium chitosan as a novel vaccine carrier for intramuscular immunization.J Biomater Appl. 2017 Nov;32(5):677-685. doi: 10.1177/0885328217735221. Epub 2017 Oct 9. J Biomater Appl. 2017. PMID: 28992775
-
In vivo investigation of twin-screw extruded lipid implants for vaccine delivery.Eur J Pharm Biopharm. 2014 Jul;87(2):338-46. doi: 10.1016/j.ejpb.2014.02.014. Epub 2014 Mar 4. Eur J Pharm Biopharm. 2014. PMID: 24607791
-
Chitosan-based particulate systems for the delivery of mucosal vaccines against infectious diseases.Int J Biol Macromol. 2018 Apr 15;110:54-64. doi: 10.1016/j.ijbiomac.2017.10.101. Epub 2017 Oct 18. Int J Biol Macromol. 2018. PMID: 29054527 Review.
Cited by
-
Nucleic Acid Delivery from Granular Hydrogels.Adv Healthc Mater. 2022 Feb;11(3):e2101867. doi: 10.1002/adhm.202101867. Epub 2021 Nov 23. Adv Healthc Mater. 2022. PMID: 34742164 Free PMC article.
-
Transitional Insight into the RNA-Based Oligonucleotides in Cancer Treatment.Appl Biochem Biotechnol. 2024 Mar;196(3):1685-1711. doi: 10.1007/s12010-023-04597-5. Epub 2023 Jul 4. Appl Biochem Biotechnol. 2024. PMID: 37402038 Review.
-
Engineering Smart Targeting Nanovesicles and Their Combination with Hydrogels for Controlled Drug Delivery.Pharmaceutics. 2020 Sep 7;12(9):849. doi: 10.3390/pharmaceutics12090849. Pharmaceutics. 2020. PMID: 32906833 Free PMC article. Review.
-
mRNA vaccines: Past, present, future.Asian J Pharm Sci. 2022 Jul;17(4):491-522. doi: 10.1016/j.ajps.2022.05.003. Epub 2022 Jun 30. Asian J Pharm Sci. 2022. PMID: 36105317 Free PMC article. Review.
-
Improved Humoral Immunity and Protection against Influenza Virus Infection with a 3d Porous Biomaterial Vaccine.Adv Sci (Weinh). 2023 Nov;10(31):e2302248. doi: 10.1002/advs.202302248. Epub 2023 Sep 26. Adv Sci (Weinh). 2023. PMID: 37750461 Free PMC article.
References
-
- Ulmer JB, Mason PW, Geall A, Mandl CW, Vaccine 2012, 30, 4414. - PubMed
-
- Siegrist C-A, Vaccine Immunology, in Vaccines 6th ed. (Eds: Plotkin SA, Orenstein WA, Offit PA), Elsevier; 2013, 14.
-
- Geall AJ, Mandl CW, Ulmer JB, Semin. Immunol 2013, 25, 152. - PubMed
-
- Yin H, Kanasty RL, Eltoukhy AA, Vegas AJ, Dorkin JR, Anderson DG, Nat. Rev. Genet 2014, 15, 541. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

