Transcriptional and functional diversity of human macrophage repolarization
- PMID: 30445062
- PMCID: PMC6451869
- DOI: 10.1016/j.jaci.2018.10.046
Transcriptional and functional diversity of human macrophage repolarization
Abstract
Background: Macrophage plasticity allows cells to adopt different phenotypes, a property with important implications in disorders such as cystic fibrosis (CF) and asthma.
Objective: We sought to examine the transcriptional and functional significance of macrophage repolarization from an M1 to an M2 phenotype and assess the role of a common human genetic disorder (CF) and a prototypical allergic disease (asthma) in this transformation.
Methods: Monocyte-derived macrophages were collected from healthy subjects and patients with CF and polarized to an M2 state by using IL-4, IL-10, glucocorticoids, apoptotic PMNs, or azithromycin. We performed transcriptional profiling and pathway analysis for each stimulus. We assessed the ability of M2-repolarized macrophages to respond to LPS rechallenge and clear apoptotic neutrophils and used murine models to determine conserved functional responses to IL-4 and IL-10. We investigated whether M2 signatures were associated with alveolar macrophage phenotypes in asthmatic patients.
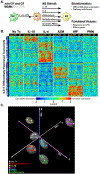
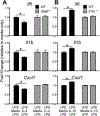
Results: We found that macrophages exhibit highly diverse responses to distinct M2-polarizing stimuli. Specifically, IL-10 activated proinflammatory pathways and abrogated LPS tolerance, allowing rapid restoration of LPS responsiveness. In contrast, IL-4 enhanced LPS tolerance, dampening proinflammatory responses after repeat LPS challenge. A common theme observed across all M2 stimuli was suppression of interferon-associated pathways. We found that CF macrophages had intact reparative and transcriptional responses, suggesting that macrophage contributions to CF-related lung disease are primarily shaped by their environment. Finally, we leveraged in vitro-derived signatures to show that allergen provocation induces distinct M2 state transcriptional patterns in alveolar macrophages.
Conclusion: Our findings highlight the diversity of macrophage polarization, attribute functional consequences to different M2 stimuli, and provide a framework to phenotype macrophages in disease states.
Keywords: Macrophage; asthma; cystic fibrosis; efferocytosis; microarray; pathway; polarization; tolerance.
Copyright © 2018 American Academy of Allergy, Asthma & Immunology. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Figures




Comment in
-
Macrophage polarization: Reaching across the aisle?J Allergy Clin Immunol. 2019 Apr;143(4):1348-1350. doi: 10.1016/j.jaci.2018.12.995. Epub 2019 Jan 9. J Allergy Clin Immunol. 2019. PMID: 30639344 No abstract available.
Similar articles
-
Azithromycin reduces exaggerated cytokine production by M1 alveolar macrophages in cystic fibrosis.Am J Respir Cell Mol Biol. 2009 Nov;41(5):590-602. doi: 10.1165/rcmb.2008-0155OC. Epub 2009 Feb 24. Am J Respir Cell Mol Biol. 2009. PMID: 19244203
-
CFTR-dependent defect in alternatively-activated macrophages in cystic fibrosis.J Cyst Fibros. 2017 Jul;16(4):475-482. doi: 10.1016/j.jcf.2017.03.011. Epub 2017 Apr 17. J Cyst Fibros. 2017. PMID: 28428011
-
ATP/P2X7r axis mediates the pathological process of allergic asthma by inducing M2 polarization of alveolar macrophages.Exp Cell Res. 2020 Jan 1;386(1):111708. doi: 10.1016/j.yexcr.2019.111708. Epub 2019 Nov 1. Exp Cell Res. 2020. PMID: 31682811
-
Macrophage plasticity, polarization, and function in health and disease.J Cell Physiol. 2018 Sep;233(9):6425-6440. doi: 10.1002/jcp.26429. Epub 2018 Mar 1. J Cell Physiol. 2018. PMID: 29319160 Review.
-
Macrophages: The Good, the Bad, and the Gluttony.Front Immunol. 2021 Aug 12;12:708186. doi: 10.3389/fimmu.2021.708186. eCollection 2021. Front Immunol. 2021. PMID: 34456917 Free PMC article. Review.
Cited by
-
Efferocytosis potentiates the expression of arachidonate 15-lipoxygenase (ALOX15) in alternatively activated human macrophages through LXR activation.Cell Death Differ. 2021 Apr;28(4):1301-1316. doi: 10.1038/s41418-020-00652-4. Epub 2020 Nov 11. Cell Death Differ. 2021. PMID: 33177619 Free PMC article.
-
New Insights into the Link Between Melanoma and Obesity.Adv Exp Med Biol. 2024;1460:851-867. doi: 10.1007/978-3-031-63657-8_28. Adv Exp Med Biol. 2024. PMID: 39287874 Review.
-
More Than Suppression: Glucocorticoid Action on Monocytes and Macrophages.Front Immunol. 2019 Aug 27;10:2028. doi: 10.3389/fimmu.2019.02028. eCollection 2019. Front Immunol. 2019. PMID: 31507614 Free PMC article. Review.
-
The protective effect of the TSPO ligands 2,4-Di-Cl-MGV-1, CB86, and CB204 against LPS-induced M1 pro-inflammatory activation of microglia.Brain Behav Immun Health. 2020 May 21;5:100083. doi: 10.1016/j.bbih.2020.100083. eCollection 2020 May. Brain Behav Immun Health. 2020. PMID: 34589858 Free PMC article.
-
The anti-inflammatory activity of 2-iminothiazolidines: evidence for macrophage repolarization.Inflammopharmacology. 2022 Dec;30(6):2427-2439. doi: 10.1007/s10787-022-01084-x. Epub 2022 Oct 22. Inflammopharmacology. 2022. PMID: 36273108
References
-
- Gordon S Alternative activation of macrophages. Nat Rev Immunol 2003; 3:23–35. - PubMed
-
- Benoit M, Desnues B, Mege JL. Macrophage polarization in bacterial infections. J Immunol 2008; 181:3733–9. - PubMed
-
- Gordon S, Taylor PR. Monocyte and macrophage heterogeneity. Nat Rev Immunol 2005; 5:953–64. - PubMed
-
- Mantovani A, Sica A, Locati M. Macrophage polarization comes of age. Immunity 2005; 23:344–6. - PubMed
-
- Vallelian F, Schaer CA, Kaempfer T, Gehrig P, Duerst E, Schoedon G, et al. Glucocorticoid treatment skews human monocyte differentiation into a hemoglobin-clearance phenotype with enhanced heme-iron recycling and antioxidant capacity. Blood 2010; 116:5347–56. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

