Surface mechanomyography and electromyography provide non-invasive indices of inspiratory muscle force and activation in healthy subjects
- PMID: 30446712
- PMCID: PMC6240075
- DOI: 10.1038/s41598-018-35024-z
Surface mechanomyography and electromyography provide non-invasive indices of inspiratory muscle force and activation in healthy subjects
Abstract
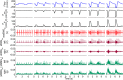
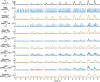
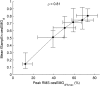
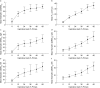
The current gold standard assessment of human inspiratory muscle function involves using invasive measures of transdiaphragmatic pressure (Pdi) or crural diaphragm electromyography (oesEMGdi). Mechanomyography is a non-invasive measure of muscle vibration associated with muscle contraction. Surface electromyogram and mechanomyogram, recorded transcutaneously using sensors placed over the lower intercostal spaces (sEMGlic and sMMGlic respectively), have been proposed to provide non-invasive indices of inspiratory muscle activation, but have not been directly compared to gold standard Pdi and oesEMGdi measures during voluntary respiratory manoeuvres. To validate the non-invasive techniques, the relationships between Pdi and sMMGlic, and between oesEMGdi and sEMGlic were measured simultaneously in 12 healthy subjects during an incremental inspiratory threshold loading protocol. Myographic signals were analysed using fixed sample entropy (fSampEn), which is less influenced by cardiac artefacts than conventional root mean square. Strong correlations were observed between: mean Pdi and mean fSampEn |sMMGlic| (left, 0.76; right, 0.81), the time-integrals of the Pdi and fSampEn |sMMGlic| (left, 0.78; right, 0.83), and mean fSampEn oesEMGdi and mean fSampEn sEMGlic (left, 0.84; right, 0.83). These findings suggest that sMMGlic and sEMGlic could provide useful non-invasive alternatives to Pdi and oesEMGdi for the assessment of inspiratory muscle function in health and disease.
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
Performance Evaluation of Fixed Sample Entropy in Myographic Signals for Inspiratory Muscle Activity Estimation.Entropy (Basel). 2019 Feb 15;21(2):183. doi: 10.3390/e21020183. Entropy (Basel). 2019. PMID: 33266898 Free PMC article.
-
Noninvasive Assessment of Neuromechanical Coupling and Mechanical Efficiency of Parasternal Intercostal Muscle during Inspiratory Threshold Loading.Sensors (Basel). 2021 Mar 4;21(5):1781. doi: 10.3390/s21051781. Sensors (Basel). 2021. PMID: 33806463 Free PMC article.
-
Assessment of Inspiratory Muscle Activation using Surface Diaphragm Mechanomyography and Crural Diaphragm Electromyography.Annu Int Conf IEEE Eng Med Biol Soc. 2018 Jul;2018:3342-3345. doi: 10.1109/EMBC.2018.8513046. Annu Int Conf IEEE Eng Med Biol Soc. 2018. PMID: 30441104
-
Clinical assessment of the respiratory muscles.Phys Ther. 1995 Nov;75(11):983-95. doi: 10.1093/ptj/75.11.983. Phys Ther. 1995. PMID: 7480128 Review.
-
Assessment of diaphragm function.Chest Surg Clin N Am. 1998 May;8(2):225-36. Chest Surg Clin N Am. 1998. PMID: 9619303 Review.
Cited by
-
Chest Movement and Respiratory Volume both Contribute to Thoracic Bioimpedance during Loaded Breathing.Sci Rep. 2019 Dec 27;9(1):20232. doi: 10.1038/s41598-019-56588-4. Sci Rep. 2019. PMID: 31882841 Free PMC article.
-
Artefact Detection in Impedance Pneumography Signals: A Machine Learning Approach.Sensors (Basel). 2021 Apr 8;21(8):2613. doi: 10.3390/s21082613. Sensors (Basel). 2021. PMID: 33917824 Free PMC article.
-
Performance Evaluation of Fixed Sample Entropy in Myographic Signals for Inspiratory Muscle Activity Estimation.Entropy (Basel). 2019 Feb 15;21(2):183. doi: 10.3390/e21020183. Entropy (Basel). 2019. PMID: 33266898 Free PMC article.
-
Noninvasive Assessment of Neuromechanical Coupling and Mechanical Efficiency of Parasternal Intercostal Muscle during Inspiratory Threshold Loading.Sensors (Basel). 2021 Mar 4;21(5):1781. doi: 10.3390/s21051781. Sensors (Basel). 2021. PMID: 33806463 Free PMC article.
-
Myths and methodologies: Invasive and non-invasive assessment of respiratory muscle activity in humans.Exp Physiol. 2025 Aug;110(8):1038-1048. doi: 10.1113/EP091526. Epub 2025 Mar 27. Exp Physiol. 2025. PMID: 40150883 Free PMC article. Review.
References
-
- American Thoracic Society/European Respiratory Society. ATS/ERS Statement on respiratory muscle testing. Am. J. Respir. Crit. Care Med. 166, 518–624 (2002). - PubMed
-
- Sharp, J. T. & Hyatt, R. E. Mechanical and electrical properties of respiratory muscles. In Handbook of Physiology, The Respiratory System, Mechanics of Breathing 389–414, 10.1002/cphy.cp030323 (1986).
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

