Large-scale conformational changes and redistribution of surface negative charge upon sugar binding dictate the fidelity of phosphorylation in Vibrio cholerae fructokinase
- PMID: 30446722
- PMCID: PMC6240065
- DOI: 10.1038/s41598-018-35236-3
Large-scale conformational changes and redistribution of surface negative charge upon sugar binding dictate the fidelity of phosphorylation in Vibrio cholerae fructokinase
Abstract
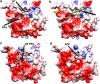
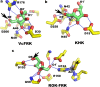
Fructokinase (FRK) catalyzes the first step of fructose metabolism i.e., D-fructose to D-fructose-6-phosphate (F6P), however, the mechanistic insights of this reaction are elusive yet. Here we demonstrate that the putative Vibrio cholerae fructokinase (VcFRK) exhibit strong fructose-6-kinase activity allosterically modulated by K+/Cs+. We have determined the crystal structures of apo-VcFRK and its complex with fructose, fructose-ADP-Ca2+, fructose-ADP-Ca2+-BeF3-. Collectively, we propose the catalytic mechanism and allosteric activation of VcFRK in atomistic details explaining why K+/Cs+ are better activator than Na+. Structural results suggest that apo VcFRK allows entry of fructose in the active site, sequester it through several conserved H-bonds and attains a closed form through large scale conformational changes. A double mutant (H108C/T261C-VcFRK), that arrests the closed form but unable to reopen for F6P release, is catalytically impotent highlighting the essentiality of this conformational change. Negative charge accumulation around ATP upon fructose binding, is presumed to redirect the γ-phosphate towards fructose for efficient phosphotransfer. Reduced phosphotransfer rate of the mutants E205Q and E110Q supports this view. Atomic resolution structure of VcFRK-fructose-ADP-Ca2+-BeF3-, reported first time for any sugar kinase, suggests that BeF3- moiety alongwith R176, Ca2+ and 'anion hole' limit the conformational space for γ-phosphate favoring in-line phospho-transfer.
Conflict of interest statement
The authors declare no competing interests.
Figures





References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

