Essential cGMP Signaling in Toxoplasma Is Initiated by a Hybrid P-Type ATPase-Guanylate Cyclase
- PMID: 30449726
- PMCID: PMC6292738
- DOI: 10.1016/j.chom.2018.10.015
Essential cGMP Signaling in Toxoplasma Is Initiated by a Hybrid P-Type ATPase-Guanylate Cyclase
Abstract
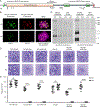
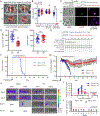
Apicomplexan parasites rely on cyclic nucleotide-dependent kinases for host cell infection, yet the mechanisms that control their activation remain unknown. Here we show that an apically localized guanylate cyclase (GC) controls microneme secretion and lytic growth in the model apicomplexan Toxoplasma gondii. Cell-permeable cGMP reversed the block in microneme secretion seen in a knockdown of TgGC, linking its function to production of cGMP. TgGC possesses an N-terminal P-type ATPase domain fused to a C-terminal heterodimeric guanylate cyclase domain, an architecture found only in Apicomplexa and related protists. Complementation with a panel of mutants revealed a critical requirement for the P-type ATPase domain for maximum GC function. We further demonstrate that knockdown of TgGC in vivo protects mice from lethal infection by blocking parasite expansion and dissemination. Collectively, this work demonstrates that cGMP-mediated signaling in Toxoplasma relies on a multi-domain architecture, which may serve a conserved role in related parasites.
Keywords: apicomplexan parasite; auxin-inducible degron; calcium; cyclic nucleotides; egress; host-pathogen interaction; invasion; regulated protein stability; secretion; signaling.
Copyright © 2018 Elsevier Inc. All rights reserved.
Figures




References
-
- Baker DA, Drought LG, Flueck C, Nofal SD, Patel A, Penzo M, and Walker EM (2017). Cyclic nucleotide signalling in malaria parasites. Open Biol 7, 10.1098/rsob.170213. - DOI - PMC - PubMed
-
- Baker DA, and Kelly JM (2004). Structure, function and evolution of microbial adenylyl and guanylyl cyclases. Mol Microbiol 52, 1229–42. - PubMed
-
- Bastin P, Bagherzadeh Z, Matthews KR, and Gull K (1996). A novel epitope tag system to study protein targeting and organelle biogenesis in Trypanosoma brucei. Molec Biochem Parasitol 77, 235–239. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

