Increased urokinase and consumption of α2 -antiplasmin as an explanation for the loss of benefit of tranexamic acid after treatment delay
- PMID: 30451372
- PMCID: PMC6334274
- DOI: 10.1111/jth.14338
Increased urokinase and consumption of α2 -antiplasmin as an explanation for the loss of benefit of tranexamic acid after treatment delay
Abstract
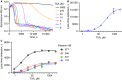
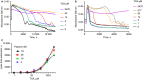
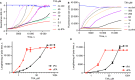
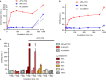
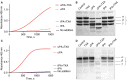
Essentials Delayed treatment with tranexamic acid results in loss of efficacy and poor outcomes. Increasing urokinase activity may account for adverse effects of late tranexamic acid treatment. Urokinase + tranexamic acid produces plasmin in plasma or blood and disrupts clotting. α2 -Antiplasmin consumption with ongoing fibrinolysis increases plasmin-induced coagulopathy. SUMMARY: Background Tranexamic acid (TXA) is an effective antifibrinolytic agent with a proven safety record. However, large clinical trials show TXA becomes ineffective or harmful if treatment is delayed beyond 3 h. The mechanism is unknown but urokinase plasminogen activator (uPA) has been implicated. Methods Inhibitory mechanisms of TXA were explored in a variety of clot lysis systems using plasma and whole blood. Lysis by tissue plasminogen activator (tPA), uPA and plasmin were investigated. Coagulopathy was investigated using ROTEM and activated partial thromboplastin time (APTT). Results IC50 values for antifibrinolytic activity of TXA varied from < 10 to > 1000 μmol L-1 depending on the system, but good fibrin protection was observed in the presence of tPA, uPA and plasmin. However, in plasma or blood, active plasmin was generated by TXA + uPA (but not tPA) and coagulopathy developed leading to no or poor clot formation. The extent of coagulopathy was sensitive to available α2 -antiplasmin. No clot formed with plasma containing 40% normal α2 -antiplasmin after short incubation with TXA + uPA. Adding purified α2 -antiplasmin progressively restored clotting. Plasmin could be inhibited by aprotinin, IC50 = 530 nmol L-1 , in plasma. Conclusions Tranexamic acid protects fibrin but stimulates uPA activity and slows inhibition of plasmin by α2 -antiplasmin. Plasmin proteolytic activity digests fibrinogen and disrupts coagulation, exacerbated when α2 -antiplasmin is consumed by ongoing fibrinolysis. Additional direct inhibition of plasmin by aprotinin may prevent development of coagulopathy and extend the useful time window of TXA treatment.
Keywords: alpha-2-antiplasmin; fibrinolysis; hemorrhage; tranexamic acid; urokinase type plasminogen activator.
© 2018 Crown copyright. Journal of Thrombosis and Haemostasis © 2018 International Society on Thrombosis and Haemostasis. This article is published with the permission of the Controller of HMSO and the Queen\u2019s Printer for Scotland.
Figures

References
-
- Ng W, Jerath A, Wasowicz M. Tranexamic acid: a clinical review. Anaesthesiol Intensive Ther 2015; 47: 339–50. - PubMed
-
- Tengborn L, Blomback M, Berntorp E. Tranexamic acid – an old drug still going strong and making a revival. Thromb Res 2015; 135: 231–42. - PubMed
-
- Longstaff C, Kolev K. Basic mechanisms and regulation of fibrinolysis. J Thromb Haemost 2015; 13(Suppl. 1): S98–105. - PubMed
-
- Silva MM, Thelwell C, Williams SC, Longstaff C. Regulation of fibrinolysis by C‐terminal lysines operates through plasminogen and plasmin but not tissue plasminogen activator (tPA). J Thromb Haemost 2012; 10: 2354–60. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

