Neutropenia in Barth syndrome: characteristics, risks, and management
- PMID: 30451719
- PMCID: PMC6392059
- DOI: 10.1097/MOH.0000000000000472
Neutropenia in Barth syndrome: characteristics, risks, and management
Abstract
Purpose of review: Barth syndrome (BTHS) is an X-linked disease characterized by defective remodeling of phospholipid side chains in mitochondrial membranes. Major features include neutropenia, dilated cardiomyopathy, motor delay and proximal myopathy, feeding problems, and constitutional growth delay. We conducted this review of neutropenia in BTHS to aid in the diagnosis of this disease, and to improve understanding of both the consequences of neutropenia and the benefits of treatment with granulocyte colony-stimulating factor (G-CSF).
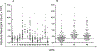
Recent findings: In 88 patients with BTHS, neutropenia, that is, at least one count below 1.5 × 10/l, was detected in 74 (84%) and 44% had severe chronic neutropenia, with multiple counts below 0.5 × 10/l. The pattern of neutropenia varied between intermittent and unpredictable, chronic and severe, or cyclical with mathematically regular oscillations. Monocytosis, that is, monocytes more than 1.0 × 10/l, was observed at least once in 64 of 85 (75%) patients. G-CSF was administered to 39 of 88 patients (44%). Weekly average G-CSF doses ranged from 0.12 to 10.92 μg/kg/day (mean 1.16 μg/kg/day, median 1.16 μg/kg/day). Antibiotic prophylaxis was additionally employed in 21 of 26 neutropenic patients. Pretreatment bone marrow evaluations predominantly showed reduced myeloid maturation which normalized on G-CSF therapy in seven of 13 examined. Consistent clinical improvement, with reduced signs and symptoms of infections, was observed in response to prophylactic G-CSF ± prophylactic antibiotics. However, despite G-CSF and antibiotics, one adult patient died with multiple infections related to indwelling medical devices and gastrostomy site infection after 15.5 years on G-CSF and a pediatric patient required gastrostomy removal for recurrent abdominal wall cellulitis.
Summary: BTHS should be considered in any men with neutropenia accompanied by any of the characteristic features of this syndrome. Prophylaxis with G-CSF ± antibiotics prevents serious bacterial infections in the more severe neutropenic patients although infections remain a threat even in patients who are very compliant with therapy, especially in those with indwelling devices.
Figures
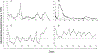

References
-
- Barth PG, Scholte HR, Berden JA, Vanderkleivanmoorsel JM, Luythouwen IEM, Vantveerkorthof ET, et al. An X-linked mitochondrial disease affecting cardiac muscle, skeletal muscle and neutrophil leucocytes. J Neurol Sci. 1983;62(1–3):327–355. - PubMed
-
- Barth PG, Wanders RJA, Vreken P. X-linked cardioskeletal myopathy and neutropenia (Barth syndrome) - MIM 302060. J Pediatr. 1999;135(3):273–276. - PubMed
-
- Bione S, Dadamo P, Maestrini E, et al. A novel X-linked gene, G4.5. is responsible for Barth syndrome. Nat Genet. 1996;12(4):385–389. - PubMed
-
- Kuijpers TW, Maianski NA, Tool ATJ, et al. Neutrophils in Barth syndrome (BTHS) avidly bind annexin-V in the absence of apoptosis. Blood. 2004;103(10):3915–3923. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

