Ruxolitinib in GvHD (RIG) study: a multicenter, randomized phase 2 trial to determine the response rate of Ruxolitinib and best available treatment (BAT) versus BAT in steroid-refractory acute graft-versus-host disease (aGvHD) (NCT02396628)
- PMID: 30453910
- PMCID: PMC6245867
- DOI: 10.1186/s12885-018-5045-7
Ruxolitinib in GvHD (RIG) study: a multicenter, randomized phase 2 trial to determine the response rate of Ruxolitinib and best available treatment (BAT) versus BAT in steroid-refractory acute graft-versus-host disease (aGvHD) (NCT02396628)
Abstract
Background: Graft-versus-Host Disease (GvHD) causes significant morbidity and mortality in patients after allogeneic stem cell transplantation. Donor T-cells cause inflammation and tissue damage in GvHD target organs such as liver, gut and skin. Cytokine receptor associated kinases JAK1 and JAK2 are critical for inflammatory cytokine response in GvHD. Ruxolitinib is a small molecule inhibitor of JAK1 and JAK2. Preliminary data indicated substantial clinical activity in patients with steroid-refractory (SR) acute and chronic GvHD.
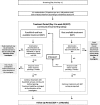
Methods: The RIG-study is an investigator-initiated open-label, multicenter, prospective randomized controlled two-arm phase 2 study, comparing the efficacy of ruxolitinib and best available treatment (BAT) versus BAT in steroid-refractory acute GvHD (SR-aGvHD). Patients with acute skin, intestinal or liver GvHD > grade 1 and failure of previous treatment are eligible. The trial aims to include 160 patients who will be randomized in a 1:1 ratio and stratified by GvHD grade (≤ grade 3 versus grade 4) and number of previous immunosuppressive treatments (≤ 3 versus ≥4). The primary endpoint is the overall response rate at day 28, defined as: Improvement of at least one stage in the severity of acute GvHD in one organ without deterioration in any other organ, or disappearance of any GvHD signs from all organs without requirement for new systemic immunosuppressive treatment. Secondary objectives include time to response, overall survival, event-free survival, non-relapse mortality (NRM), failure-free survival, graft failure rates, quality of life and changes in serum levels of pro-inflammatory cytokines and GvHD-related biomarkers.
Discussion: This randomized prospective trial will provide further evidence if the retrospectively collected data demonstrating activity of ruxolitinib for SR-aGvHD can be reproduced. A major advantage of ruxolitinib might be the limited and predictable toxicity profile compared to other immunosuppressive therapies that mainly includes viral reactivation and cytopenias. This trial will establish candidate biomarkers to predict and monitor responses to ruxolitinib. As a next step ruxolitinib might be tested upfront against steroids or in a preemptive manner to prevent GvHD to occur.
Trial registration: NCT02396628 (registration date 17.07.2015); DRKS00007939 (registration date 26.03.2015).
Keywords: Biomarkers; Inflammatory cytokines; JAK inhibitor; Ruxolitinib; Steroid-refractory GvHD.
Conflict of interest statement
Ethics approval and consent to participate
The study protocol has been approved by the German Federal Institute for Drugs and Medicine Devices and by the leading independent Ethics Committee of the University of Freiburg (EK 290/16). The study will be conducted in accordance with the ethical principles of the Declaration of Helsinki, the ICH-GCP Guidelines, the EU Clinical Trial Directive (2001/20/EG) and applicable regulatory requirements. Patient’s written consent to participate in this clinical trial is obtained prior to any study-specific procedures. The RIG-study has been registered in the
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
-
- Xhaard A, et al. Steroid-refractory acute GVHD: lack of long-term improved survival using new generation Anticytokine treatment. Biol Blood Marrow Transplant. 2011. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous