The Italian autism network (ITAN): a resource for molecular genetics and biomarker investigations
- PMID: 30463616
- PMCID: PMC6247619
- DOI: 10.1186/s12888-018-1937-y
The Italian autism network (ITAN): a resource for molecular genetics and biomarker investigations
Abstract
Background: A substantial genetic component accounts for Autism Spectrum Disorders (ASD) aetiology, with some rare and common genetic risk factors recently identified. Large collections of DNAs from thoroughly characterized ASD families are an essential step to confirm genetic risk factors, identify new variants and investigate genotype-phenotype correlations. The Italian Autism Network aimed at constituting a clinical database and a biorepository of samples derived from ASD subjects and first-degree relatives extensively and consistently characterized by child psychiatry centers in Italy.
Methods: The study was approved by the ethical committee of the University of Verona, the coordinating site, and by the local ethical committees of each recruiting site. Certified staff was specifically trained at each site for the overall study conduct, for clinical protocol administration and handling of biological material. A centralized database was developed to collect clinical assessment and medical records from each recruiting site. Children were eligible for recruitment based on the following inclusion criteria: age 4-18 years, at least one parent or legal guardian giving voluntary written consent, meeting DSM-IV criteria for Autistic Disorder or Asperger's Disorder or Pervasive Developmental Disorder NOS. Affected individuals were assessed by full psychiatric, neurological and physical examination, evaluation with ADI-R and ADOS scales, cognitive assessment with Wechsler Intelligence Scale for Children or Preschool and Primary, Leiter International Performance Scale or Griffiths Mental Developmental Scale. Additional evaluations included language assessment, the Krug Asperger's Disorder Index, and instrumental examination such as EEG and structural MRI. DNA, RNA and plasma were collected from eligible individuals and relatives. A central laboratory was established to host the biorepository, perform DNA and RNA extraction and lymphocytes immortalisation.
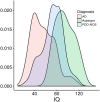
Discussion: The study has led to an extensive collection of biological samples associated with standardised clinical assessments from a network of expert clinicians and psychologists. Eighteen sites have received ADI/ADOS training, thirteen of which have been actively recruiting. The clinical database currently includes information on 812 individuals from 249 families, and the biorepository has samples for 98% of the subjects. This effort has generated a highly valuable resource for conducting clinical and genetic research of ASD, amenable to further expansion.
Keywords: Autism Spectrum disorders; Biomarkers; Biorepository; Genetics.
Conflict of interest statement
Ethics approval and consent to participate
The study protocol was in first instance approved by the Verona Hospital Ethical Review board (study protocol AUT-SFK001, CE1419), and subsequently by following Ethical Review Committees of each recruiting site:
Comitato Etico per la Sperimentazione, Azienda Ospedaliera di Verona, Verona, Italy
Comitato Etico dell’Azienda Ospedaliera Universitaria, Seconda Università degli studi di Napoli, Napoli, Italy
Comitato Etico di Area Vasta Romagna ed. IRST, Meldola (FC), Italy
Comitato Etico Indipendente locale, Azienda Ospedaliera “Ospedale Policlinico Consorziale” di Bari, Bari, Italy
Comitato Etico Azienda Ospedaliera Spedali Civili di Brescia, Brescia, Italy
Comitato Etico Azienda Ospedaliero Universitaria di Cagliari, Cagliari, Italy
Comitato Etico IRCCS Associazione Oasi Maria SS. ONLUS, Troina (EN), Italy
Comitato Etico Indipendente PTV Azienda Ospedaliera Universitaria, Policlinico di Tor Vergata, Roma (Italy)
Comitato Etico Associazione La Nostra Famiglia IRCCS E. Medea, Bosisio Parini (LC), Italy
Comitato Etico di Fondazione Stella Maris, Calambrone (PI), Italy
Comitato Etico Indipendente Servizio Sanitario Regionale Emilia Romagna, Azienda Unitaria Sanitaria Locale di Bologna, Italy
Comitato Etico per la Sperimentazione Azienda Ospedaliera di Padova, Padova, Italy
Comitato Etico Azienda Sanitaria Provinciale Catania, Catania, Italy
All adult subjects participating in this project gave their written consent (or the consent for their chidren) to donate biological samples and clinical and demographic information to participate in this study; assent to participate to this study from the children was obtained whenever obtainable. A multidisciplinary Scientific and Ethical Committee of the Fondazione SmithKline, “Area Ricerca Autismo”, i) created the protocol; ii) ensured that the use of the samples and clinical database was consistent with what consented by the research participants; iii) ensured that research was conducted with sound methodology and by accredited institutions.
Consent for publication
Not applicable.
Competing interests
PM is an employee of UCB Pharma (Belgium); ER is an employee of Takeda (USA); TKT is an employee of F. Hoffman-La Roche Ltd. (Switzerland); PR is an employee of ViiV Healthcare. PM, ED, LDR and ER were employees of GSK (Italy) during the design and launch of the clinical study. ED has received research support from Roche in the period 2016–2018. AZ has served on the advisory boards of Shire, EcuPharma and Angelini and on data safety monitoring boards of Otsuka and Lundbeck; he has received research support from Shire, Vifor, Roche, Lundbeck, Jannssen.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures

References
-
- American Psychiatric Association. Diagnostic and statistical manual of mental disorders. 5th ed. Washington, DC: American Psychiatric Association; 2013.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

