A Human Gain-of-Function STING Mutation Causes Immunodeficiency and Gammaherpesvirus-Induced Pulmonary Fibrosis in Mice
- PMID: 30463976
- PMCID: PMC6364005
- DOI: 10.1128/JVI.01806-18
A Human Gain-of-Function STING Mutation Causes Immunodeficiency and Gammaherpesvirus-Induced Pulmonary Fibrosis in Mice
Abstract
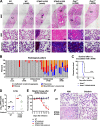
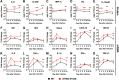
We previously generated STING N153S knock-in mice that have a human disease-associated gain-of-function mutation in STING. Patients with this mutation (STING N154S in humans) develop STING-associated vasculopathy with onset in infancy (SAVI), a severe pediatric autoinflammatory disease characterized by pulmonary fibrosis. Since this mutation promotes the upregulation of antiviral type I interferon-stimulated genes (ISGs), we hypothesized that STING N153S knock-in mice may develop more severe autoinflammatory disease in response to a virus challenge. To test this hypothesis, we infected heterozygous STING N153S mice with murine gammaherpesvirus 68 (γHV68). STING N153S mice were highly vulnerable to infection and developed pulmonary fibrosis after infection. In addition to impairing CD8+ T cell responses and humoral immunity, STING N153S also promoted the replication of γHV68 in cultured macrophages. In further support of a combined innate and adaptive immunodeficiency, γHV68 infection was more severe in Rag1-/- STING N153S mice than in Rag1-/- littermate mice, which completely lack adaptive immunity. Thus, a gain-of-function STING mutation creates a combined innate and adaptive immunodeficiency that leads to virus-induced pulmonary fibrosis.IMPORTANCE A variety of human rheumatologic disease-causing mutations have recently been identified. Some of these mutations are found in viral nucleic acid-sensing proteins, but whether viruses can influence the onset or progression of these human diseases is less well understood. One such autoinflammatory disease, called STING-associated vasculopathy with onset in infancy (SAVI), affects children and leads to severe lung disease. We generated mice with a SAVI-associated STING mutation and infected them with γHV68, a common DNA virus that is related to human Epstein-Barr virus. Mice with the human disease-causing STING mutation were more vulnerable to infection than wild-type littermate control animals. Furthermore, the STING mutant mice developed lung fibrosis similar to that of patients with SAVI. These findings reveal that a human STING mutation creates severe immunodeficiency, leading to virus-induced lung disease in mice.
Keywords: MHV68; STING; adaptive immunity; autoimmunity; gammaherpesvirus; innate immunity; pulmonary fibrosis; vasculopathy.
Copyright © 2019 American Society for Microbiology.
Figures







Similar articles
-
STING-associated vasculopathy develops independently of IRF3 in mice.J Exp Med. 2017 Nov 6;214(11):3279-3292. doi: 10.1084/jem.20171351. Epub 2017 Sep 26. J Exp Med. 2017. PMID: 28951494 Free PMC article.
-
STING-associated lung disease in mice relies on T cells but not type I interferon.J Allergy Clin Immunol. 2019 Jul;144(1):254-266.e8. doi: 10.1016/j.jaci.2019.01.044. Epub 2019 Feb 14. J Allergy Clin Immunol. 2019. PMID: 30772497 Free PMC article.
-
Hierarchy of clinical manifestations in SAVI N153S and V154M mouse models.Proc Natl Acad Sci U S A. 2019 Apr 16;116(16):7941-7950. doi: 10.1073/pnas.1818281116. Epub 2019 Apr 3. Proc Natl Acad Sci U S A. 2019. PMID: 30944222 Free PMC article.
-
Lung Inflammation in STING-Associated Vasculopathy with Onset in Infancy (SAVI).Cells. 2022 Jan 18;11(3):318. doi: 10.3390/cells11030318. Cells. 2022. PMID: 35159128 Free PMC article. Review.
-
STING-Mediated Lung Inflammation and Beyond.J Clin Immunol. 2021 Apr;41(3):501-514. doi: 10.1007/s10875-021-00974-z. Epub 2021 Feb 2. J Clin Immunol. 2021. PMID: 33532887 Review.
Cited by
-
Cytoplasmic DNAs: Sources, sensing, and roles in the development of lung inflammatory diseases and cancer.Front Immunol. 2023 Apr 12;14:1117760. doi: 10.3389/fimmu.2023.1117760. eCollection 2023. Front Immunol. 2023. PMID: 37122745 Free PMC article. Review.
-
Stimulator of Interferon Genes-Associated Vasculopathy With Onset in Infancy: A Systematic Review of Case Reports.Front Pediatr. 2020 Dec 17;8:577918. doi: 10.3389/fped.2020.577918. eCollection 2020. Front Pediatr. 2020. PMID: 33425809 Free PMC article.
-
STING trafficking as a new dimension of immune signaling.J Exp Med. 2023 Mar 6;220(3):e20220990. doi: 10.1084/jem.20220990. Epub 2023 Jan 27. J Exp Med. 2023. PMID: 36705629 Free PMC article.
-
Regulation of cGAS and STING signaling during inflammation and infection.J Biol Chem. 2023 Jul;299(7):104866. doi: 10.1016/j.jbc.2023.104866. Epub 2023 May 27. J Biol Chem. 2023. PMID: 37247757 Free PMC article. Review.
-
STING negatively regulates allogeneic T-cell responses by constraining antigen-presenting cell function.Cell Mol Immunol. 2021 Mar;18(3):632-643. doi: 10.1038/s41423-020-00611-6. Epub 2021 Jan 26. Cell Mol Immunol. 2021. PMID: 33500563 Free PMC article.
References
-
- Kobayashi H, Kobayashi CI, Nakamura-Ishizu A, Karigane D, Haeno H, Yamamoto KN, Sato T, Ohteki T, Hayakawa Y, Barber GN, Kurokawa M, Suda T, Takubo K. 2015. Bacterial c-di-GMP affects hematopoietic stem/progenitors and their niches through STING. Cell Rep 11:71–84. doi:10.1016/j.celrep.2015.02.066. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

